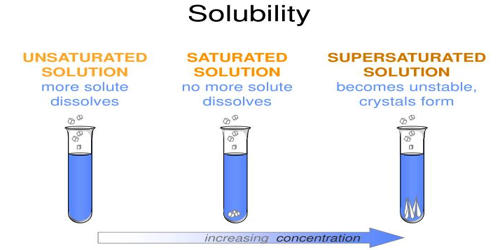
Solubility is a property referring to the ability for a given substance, the solute, to dissolve in a solvent. It is the property of a solid, liquid, or a gaseous chemical substance called solute to dissolve in a solid, liquid, or gaseous solvent. It is measured in terms of the maximum amount of solute dissolved in a solvent at equilibrium. The solubility of a substance fundamentally depends on the physical and chemical properties of the solute and solvent as well as on temperature, pressure, and presence of other chemicals (including changes to the pH) of the solution. Solubilities range widely, from infinitely soluble such as ethanol in water to poorly soluble, such as silver chloride in water. The extent of the solubility of a substance in a specific solvent is measured as the saturation concentration, where adding more solute does not increase the concentration of the solution and begins to precipitate the excess amount of solute.
Insolubility is the inability to dissolve in a solid, liquid, or gaseous solvent. The definition of solubility is the maximum quantity of solute that can dissolve in a certain quantity of solvent or quantity of solution at a specified temperature or pressure.
So, the maximum amount of solute that can dissolve in a known quantity of solvent at a certain temperature is its solubility. Most often, the solvent is a liquid, which can be a pure substance or a mixture. One may also speak of solid solution, but rarely of solution in a gas. The solubility of a substance fundamentally depends on the solvent used, as well as temperature and pressure. The solubility of a substance in a particular solvent is measured by the concentration of the saturated solution
Applications
- Solubility is of fundamental importance in a large number of scientific disciplines and practical applications, ranging from ore processing and nuclear reprocessing to the use of medicines, and the transport of pollutants.
- Solubility is often said to be one of the “characteristic properties of a substance”, which means that solubility is commonly used to describe the substance, to indicate a substance’s polarity, to help to distinguish it from other substances, and as a guide to applications of the substance.
- The solubility of a substance is useful when separating mixtures. For example, a mixture of salt (sodium chloride) and silica may be separated by dissolving the salt in water and filtering off the undissolved silica.
Factors Affecting Solubility
- Temperature – For many solids dissolved in liquid water, solubility tends to correspond with increasing temperature. As water molecules heat up, they vibrate more quickly and are better able to interact with and break apart the solute.
- Pressure – Pressure has a negligible effect on the solubility of solid and liquid solutes, but it has a strong effect on solutions with gaseous solutes.
- Polarity – A popular saying used for predicting solubility is “Like dissolves like.” This statement indicates that a solute will dissolve best in a solvent that has a similar chemical structure; the ability for a solvent to dissolve various compounds depends primarily on its polarity.