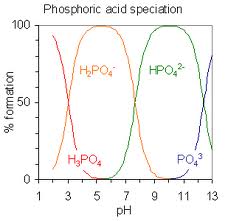
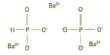
This article focus to Define and Discuss on Polyprotic Acids. Many acids contain two or more ionizable hydrogens. There usually are two in carbonic acid solution, H 2CO 3, in addition to three in phosphoric acid solution, H 3PO 4. For just about any such multiple hydrogen acid solution, the first hydrogen is usually most easily removed, and the last hydrogen is removed with the greatest difficulty. These acids usually are called polyprotic (many protons) acids.
Define and Discuss on Polyprotic Acids