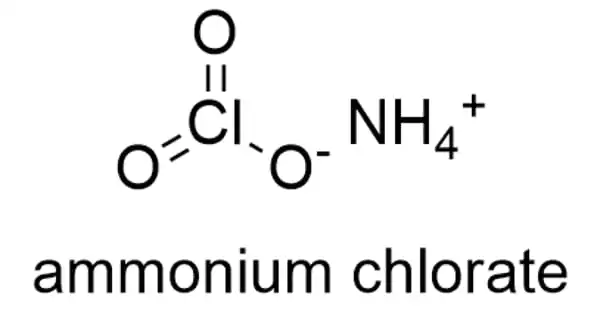
Ammonium chlorate is an inorganic chemical having the formula NH4ClO3. It’s an ionic compound made up of two polyatomic ions. In this lesson, we will look at the formula of ammonium chlorate, as well as its solubility and applications.
It is made by neutralizing chloric acid with ammonia or ammonium carbonate, or by precipitating barium, strontium, or calcium chlorates with ammonium carbonate or ammonium sulfate, yielding the corresponding carbonate or sulfate precipitate and an ammonium chlorate solution. Ammonium chlorate crystallizes in the form of tiny needles that are easily soluble in water.
Properties
Ammonium chlorate is a very unstable salt that is primarily employed in the production of explosives. Its pure form is exceedingly unstable and prone to exploding. It’s a clear, colorless crystal salt. It has a density of 2.42 g mL-1 and a melting point of 3800C. It is soluble in water and diluted alcohol solutions, but in pure alcohols it is insoluble.
- Appearance: Small colorless crystals
- Density: 2.42 g/cm3
- Melting Point: 380°C
- Molar Mass: 101.49 g/mol
- Color: White, unstable, colorless crystals or needles

Production
The bitartrate method is a manufacturing candidate that can be used if unusual chlorates are currently unavailable or must be manufactured. Warm potassium chlorate and ammonium bitartrate solutions are required. The latter can be made by combining aqueous ammonia with an excess of tartaric acid. The precipitation of ammonium chlorate will then occur as a result of a double displacement process.
Preparation
Ammounium chlorate can be made by mixing stoichiometric solutions of ammonium nitrate and sodium chlorate or ammonium sulfate and barium chlorate.
Ammonium chlorate is prepared from the neutralization of chloric acid with ammonia or ammonium carbonate:
HClO3 + NH3 → NH4ClO3
It can be prepared by reacting barium or calcium chlorate with ammonium sulfate to obtain the ammonium chlorate and barium or calcium sulfate. Calcium and barium cations form insoluble sulfates in water, thus it can be easily removed from the reaction mix.
Ca(ClO3)2 + (NH4)2SO4 → 2NH4ClO3 + CaSO4(s)
Solubility
Ammonium chlorate decomposes when heated to roughly 102 °C, releasing nitrogen, chlorine, and oxygen. It dissolves in weak aqueous alcohol but not in strong alcohol. This compound is a strong oxidant and should never be stored with combustible materials because it can readily generate explosive compositions.
At room temperature, ammonium chlorate is an extremely unstable oxidant that will break down independently, occasionally violently. This is caused by the interaction of the reducing ammonium cation and the oxidizing chlorate anion. Even solutions are often unstable. Because of the dangers associated with this salt, it should only be kept in solution when needed and should never be allowed to crystallize.
Uses
Ammonium chlorate has a fairly limited range of applications. The principal application for unstable salts is typically in the creation of explosive or chemical weapons.
Health effects/safety hazards
Ammonium chlorate is extremely hazardous. Its shipment is forbidden because to the significant risk of explosions. When exposed to light or heat, it can violently explode in concentrated solutions.