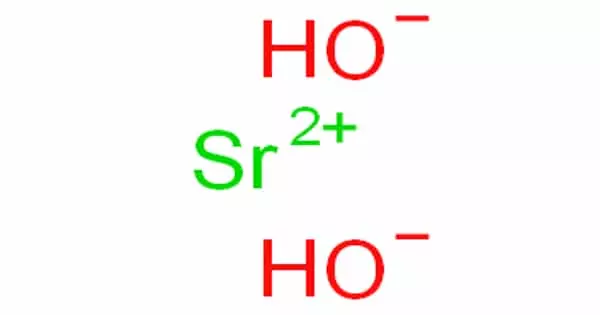
Strontium hydroxide, Sr(OH)2 is a caustic alkali composed of one strontium ion and two hydroxide ions. It is a deliquescent solid that forms a crystalline octahydrate, dissolves in water to form a decidedly alkaline solution, and is primarily used in the production of soaps and greases, as well as in the refining of beet sugar and the recovery of sugar from molasses through the formation of insoluble sucrates. It is slightly soluble in water, but the octahydrate is far more soluble.
It is made by combining a strontium salt and a strong base. It can be anhydrous, monohydrate, or octahydrate. It exists as an anhydrate with deliquescent prismatic, colorless crystals.
Properties
- Molecular Weight: 121.63
- Appearance: White Crystalline Solid
- Melting Point: 375° C (707° F)
- Boiling Point: 710° C (1,310° F)
- Density: 3.62 g/cm3
- Solubility in H2O: N/A
- Exact Mass: 121.911094
- Specific Gravity: 1.9 gm/ml

Preparation
Because Sr(OH)2 is only slightly soluble in cold water, it is simple to prepare by adding a strong base, such as NaOH or KOH, drop by drop to a solution of any soluble strontium salt, most commonly Sr(NO3)2 (strontium nitrate). Sr(OH) will form a fine white powder as it precipitates. The solution is then filtered, and the Sr(OH)2 is washed and dried with cold water.
It can be made by reacting water with the oxide, but because Sr(OH)2 is only very slightly soluble in water, it can be easily made by adding a strong base, such as NaOH or KOH, to a solution of any strontium salt, most commonly the nitrate, Sr(NO3)2.
Applications
Strontium hydroxide is primarily used in the refining of beet sugar and as a plastic stabilizer. When the chlorine from strontium chloride is undesirable, it can be used as a source of strontium ions. To form strontium carbonate, strontium hydroxide absorbs carbon dioxide from the air.
Safety
Strontium hydroxide is a severe irritant to the skin, eyes, and respiratory tract. If swallowed, it is poisonous.