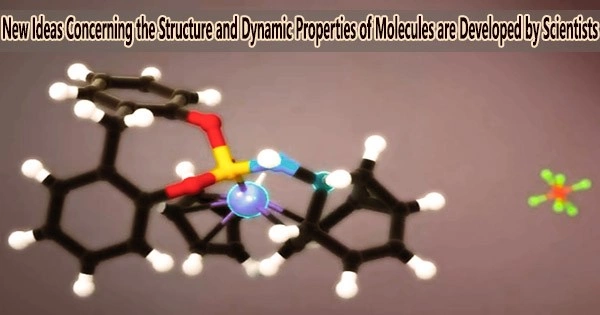
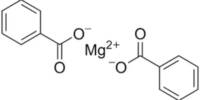
In a recent study, researchers showed that molecules made of carbon can be significantly more dynamic than previously believed.
A carbon atom can generate two mirror images of the molecule when it links four times to other groups. Because they have various biological actions, these mirror image forms are essential in medicine.
Most of the time, it is impossible to switch between these “enantiomers” because doing so would require breaking a bond, which would demand an excessive amount of energy.
The Durham University and University of York researchers showed that if the chiral center was a component of a dynamic molecular cage structure, then a straightforward rearrangement of the cage might result in the inversion of the molecule’s mirror image form.
By doing this, carbon-based stereochemistry, which is typically thought of as stable and rigid, can transform into a new paradigm in carbon-centered chirality that is dynamic, fluxional, and responsive.
The findings are published in the journal Nature Chemistry.
The molecular cage has nine carbons atoms in its structure, which are held together by a pair of carbon–carbon double bonds and a three-membered cyclopropane ring. Some of the bonds in the structure can swap places with one another spontaneously thanks to this arrangement of bonds.
The way our dynamic carbon cage interacts with other molecules and ions is fascinating. The cage adapts, giving the mirror-image structure with the best fit. We hope, in due course that this intriguing bonding concept will be found to apply in other contexts, and potentially used to underpin new applications for more dynamic molecular materials.
Dr. Paul McGonigal
Project lead investigator, Dr. Aisha Bismillah of Durham University, said, “Our dynamic carbon cages change their shape extremely quickly. They hop back and forth between their mirror image structures millions of times a second. Seeing them adapt to match changes in their environment is truly remarkable.”
By demonstrating that the preferences of the cage could be transmitted to nearby metal centers, the researchers further revealed this exceptional dynamic form of stereochemical interconversion, raising the possibility that this type of responsive chirality might find applications in catalysis and the synthesis of chiral molecules for biomedical applications.
Reflecting on the way in which these results overturn established ideas, Dr. Paul McGonigal of University of York, said, “The way our dynamic carbon cage interacts with other molecules and ions is fascinating. The cage adapts, giving the mirror-image structure with the best fit.”
“We hope, in due course that this intriguing bonding concept will be found to apply in other contexts, and potentially used to underpin new applications for more dynamic molecular materials.”