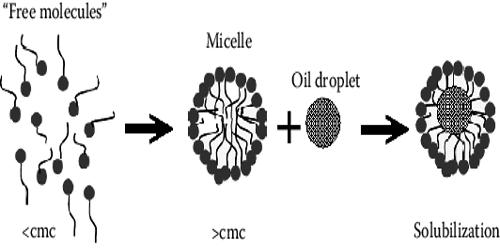
Micellar solubilization (or solubilization) is a powerful alternative for dissolving hydrophobic drugs in aqueous environments. It is the process of incorporating the solubilizate into or onto micelles. Solubilization may occur in a system consisting of a solvent, an association colloid (a colloid that forms micelles), and at least one other solubilizate.
Usage of the term
Solubilization is distinct from dissolution because the resulting fluid is a colloidal dispersion involving an association colloid. This suspension is distinct from a true solution, and the amount of the solubilizate in the micellar system can be different than the regular solubility of the solubilizate in the solvent. Therefore, the study of surfactants and their role in pharmacy is of paramount importance, especially with respect to their ability of solubilizing hydrophobic drugs.
The micelles have an important property that has exact importance in pharmacy is their ability to increase the solubility of least soluble substances in water. In non-chemical literature and in everyday language, the term “solubilization” is sometimes used in a broader meaning as “to bring to a solution or (non-sedimenting) suspension” by any means, e.g., leaching by a reaction with an acid. In this respect, solubilization can be termed as the vigorous solubility of a substance by reversed interaction with the surfactant micelles in water to form a thermodynamically stable isotropic solution with the reduced of thermodynamic activity of the material that is to be solubilized.
Application
Micellar solubilization is widely utilized, e.g. in laundry washing using detergents, in the pharmaceutical industry, for formulations of poorly soluble drugs in solution form, and in the cleanup of oil spills using dispersants.
The solubilization of phenolic compounds such as cresol, chlorocresol, chloroxylenol, and thymol with soap to produce clear solutions for use in the disinfection.
Iodine solubilized solution in non-ionic surfactant micelles (iodophors) are used in instrument sterilization.
Solubilization of drug (such as steroids and insoluble water vitamins), and necessary oils aside non-ionic surfactants (generally polysorbates or polyoxyethylene sorbitan esters of fatty acids).
Micellar systems can solubilize poorly soluble drugs and thus increase their bioavailability, they can stay in the body (blood) long enough to provide gradual accumulation in the required area, and their sizes permit them to accumulate in areas with leaky vasculature. The application of micelles in drug delivery, in order to minimize drug degradation and loss, to prevent harmful side effects, and to increase drug bioavailability, is also presented. The drug paclitaxel is highly lipophilic in nature and insoluble in water. Its current marketed form as intravenous infusion paclitaxel is solubilized by using Cremophor EL, which is a nonionic surfactant and helps in the micellar solubilization.