Introduction
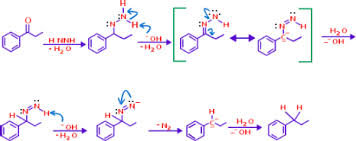
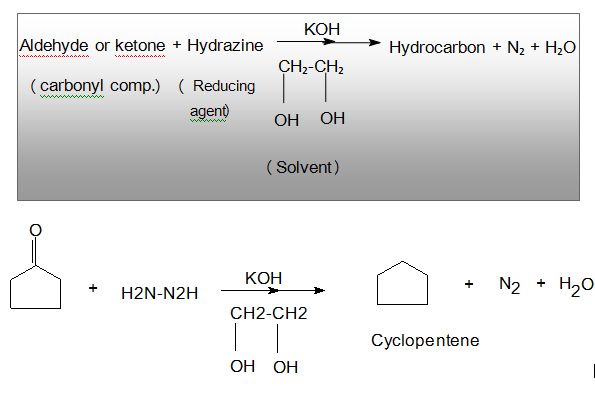
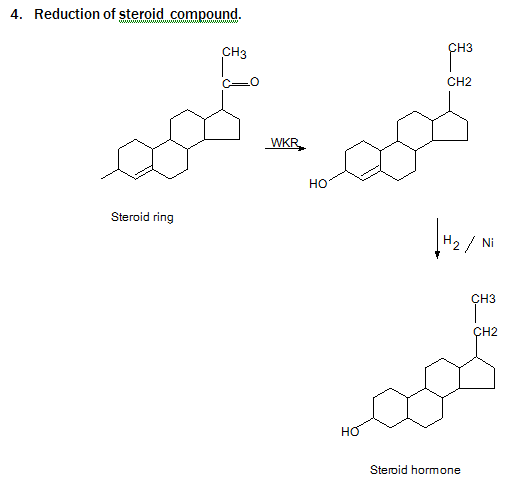
Wolf Kishner Reduction is reaction ,where Carbonyl compound are converted to hydrocarbon by the action of hydrazine ( H₂N – NH₂ ) and base (KOH),the method originally involved heating the hydrazine with sodium ethoxide in a sealed vessel at above 180* C temperature . Other bases have found equally effective. Di-ethylene glycol is usually used as solvent.
Wolf Kishner Reduction
Principle of Reaction: In this reaction ,Carbonyl compound are converted to hydrocarbon by the action of hydrazine ( H₂N – NH₂ ) and base (KOH),the method originally involved heating the hydrazine with sodium ethoxide in a sealed vessel at above 180* C temperature . Other bases have found equally effective. Di-ethylene glycol is usually used as solvent.
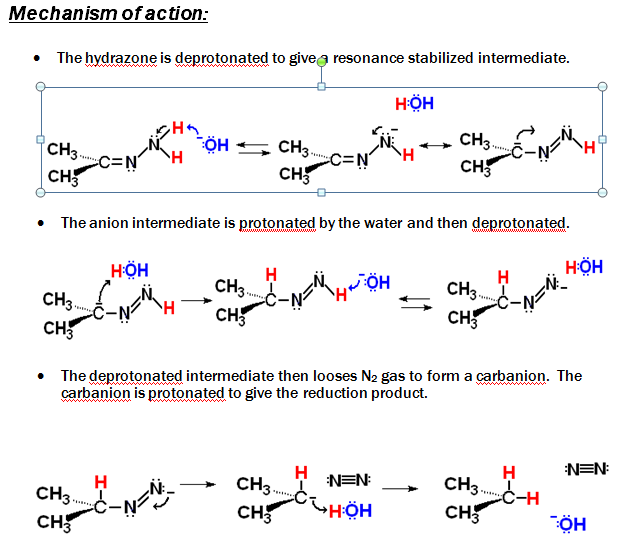
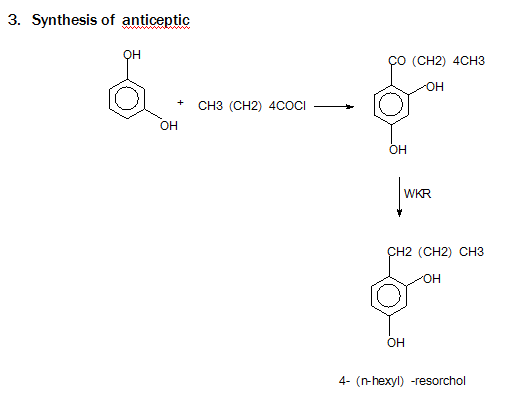
Limitation:
- This reduction is only applicable for acid-sensitive aldehyde or ketone.
- It is much more preferable for aldehyde rather then ketone because of steric hindrance due to the appearance of multigroup.
- Some side products may be obtained.