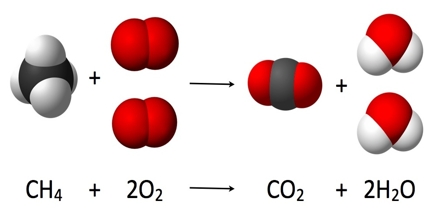
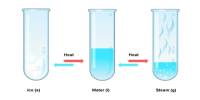
Primary objective of this lecture is to focus on understanding Chemical Reactions. For a chemical reaction to occur, bonds must be both formed and broken. Reactions occur when two or more molecules interact and the molecules change. Chemical reactions always involve energy changes. Making and breaking bonds involves energy changes. Here also focus on Exothermic reactions; hese reactions give out heat energy, Combustion is an exothermic reaction.
Understanding Chemical Reactions