According to scientists at Dana-Farber Cancer Institute and in Israel, the human immune system has a billion-year-old family of proteins employed by bacteria to fight themselves against viruses.
The findings, which were published today in the journal Science, are the latest in a growing body of evidence showing components of the human immune system evolved early in ancient forms of life, making it the most advanced disease-fighting system on the planet.
The findings reveal that the immune system absorbed already existing elements and put them to novel uses over millennia of evolution to satisfy the needs of species as biologically complex as humans.
“There has been a tremendous amount of work by researchers around the world to understand how the human immune system functions,” says the study’s senior author, Philip Kranzusch, Ph.D., of Dana-Farber. “The discovery that key parts of human immunity are shared in bacteria provides a new blueprint for research in this area.”
Your child’s immune system defends his or her body against foreign invaders. These include bacteria, viruses, and fungus, as well as poisons (chemicals made by microbes). Different organs, cells, and proteins make up the immune system, and they all operate together.
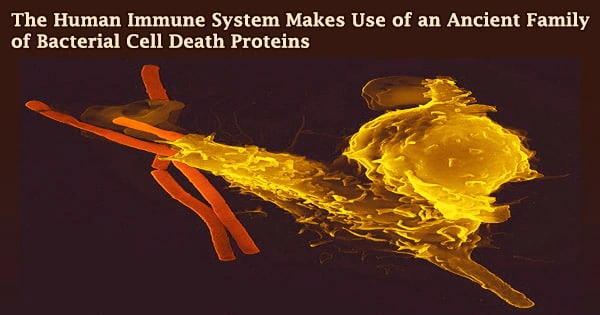
Gasdermins are the proteins at the focus of the research. Gasdermins create pores in a cell’s membrane when it gets contaminated or malignant, leading the cell to die. Inflammatory cytokines leak out the perforations, signifying the presence of infection or malignancy and causing the immune system to mobilize in defense of the body.
This mechanism, known as pyroptosis, is part of the immune system’s arsenal for eliminating damaged or infected cells. It works in tandem with the more well-known apoptosis mechanism, in which damaged or infected cells self-destruct.
There has been a tremendous amount of work by researchers around the world to understand how the human immune system functions. The discovery that key parts of human immunity are shared in bacteria provides a new blueprint for research in this area.
Philip Kranzusch
“Pyroptosis represents one of the fastest ways that the innate immune system the body’s first line of defense against pathogens responds to potential threats,” says the new study’s co-first author, Alex Johnson, Ph.D., of Dana-Farber.
Six gasdermin proteins are encoded in the human genome, and they are expressed at different amounts in different cell types. Johnson and his colleagues wanted to see if any of these proteins’ ancestors existed in bacteria for this investigation.
They had strong reason to believe they could. Kranzusch and his colleagues discovered in 2019 that bacteria gave rise to a human immunological signaling system termed cGAS-STING, which detects anomalies connected to cancer and infection.
“This and other discoveries motivated us to look for additional connections between immune-related proteins in human and bacterial cells,” Kranzusch notes.
Tanita Wein, Ph.D., Rotem Sorek, Ph.D., and colleagues at Israel’s Weizmann Institute of Science examined regions of bacterial DNA known as “antiphage defense islands,” which contain clusters of genes that protect bacteria from infection by viruses known as phages. They discovered 50 bacterial genes that were anticipated to produce proteins with a structure comparable to that of mammalian gasdermin proteins.
“I determined a series of structures of these proteins using X-ray crystallography, which confirmed at atomic detail their architectural similarity with mammalian gasdermins,” Johnson relates.
These proteins are carried by bacteria that can be found in soil, leaves, and other natural settings. (The bacterium at the center of Johnson’s research was first discovered on a Wisconsin maize plant.)
While human and bacterial gasdermins are physically similar, the bacterial versions are about half the size and serve as building blocks for membrane pores larger than those seen in humans, according to Johnson’s research. A similar process activates all of these gasdermins, but the chain of actions they initiate in human cells is significantly more comprehensive.
Viral infection in bacterial cells can cause cells to die due to perforated membranes, thereby stopping viruses in their tracks. The death of an infected cell in human cells sets off a chain of events that brings additional immune system components to bear on the infection.
“This is an example of a very primitive form of defense, which, in humans, has been adapted and expanded with regulatory systems that enable our bodies to respond to infection or cancer,” Kranzusch says.
Within the amazing intricacy of the human immune system, researchers have discovered signs of a primitive form of immunity, which can help researchers better understand how the system came to be.
“Seeing the simplest version of a machine can give you a new level of understanding of the machine as a whole,” Kranzusch remarks. “The same principle can apply to research into the immune system.”
Co-authors of the study are: Brianna Duncan-Lowey, Ph.D., of Dana-Farber; Megan Mayer, MS, of the Harvard Center for Cryo-Electron Microscopy; and Erez Yimiya, Yaara Oppenheimer-Shaanan, Ph.D., and Gil Amitai, Ph.D., of the Weizmann Institute.
Funding for the study was provided by: the Pew Biomedical Scholars Program; the Burroughs Wellcome Fund; the Mathers Foundation; the Parker Institute for Cancer Immunotherapy; the European Research Council; the Israel Science Foundation; the Ernest and Bonnie Beutler Research Program of Excellence; the Minerva Foundation and Federal German Ministry for Education and Research; the Knell Family Center for Microbiology; the Yotam project and the Weizmann Institute Sustainability and Energy Research Initiative; the Dr. Barry Sherman Institute for Medicinal Chemistry; the National Institute of Health Cancer Immunology (training grant T32CA207021); a Life Science Research Foundation postdoctoral fellowship of the Open Philanthropy Project; a Minerva Foundation postdoctoral fellowship; and a Herchel Smith Graduate Research Fellowship.