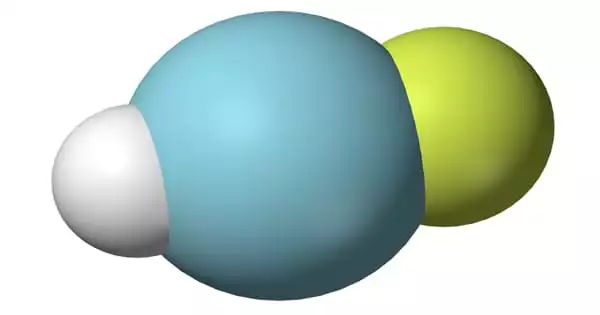
Magnesium Chloride
Definition
Magnesium chloride is a chemical element and its atomic number 12, atomic weight 24.312, symbol Mg. It is an inorganic salt (compound) that has one magnesium and two chloride ions with the chemical formula MgCl2. Its various hydrates have the formula MgCl2(H2O)x. It is used as a cathartic and in alloys. These salts are typical ionic halides, being highly soluble in water. The hydrated magnesium chloride can be extracted from brine or sea water. In North America, magnesium chloride is produced primarily from Great Salt Lake brine.

Magnesium Chloride is recognized by many medical professionals as the “Master Magnesium Compound” for both dietary and topical uses, due to its high potency and efficient action. Magnesium is undeniably therapeutic to the human body, yet much is left unsaid about the type and quality of various forms of magnesium.
Some magnesium chloride is made from solar evaporation of seawater. Anhydrous magnesium chloride is the principal precursor to magnesium metal, which is produced on a large scale. Hydrated magnesium chloride is the form most readily available. Hydrated magnesium chloride can be produced by extraction from brine or seawater. Anhydrous magnesium chloride is produced on a large scale and it is the primary precursor to magnesium metal.
Structure and Properties of Magnesium Chloride
Magnesium chloride is an inorganic compound containing the elements magnesium and chlorine. It is crystallizes in the cadmium chloride motif, which features octahedral Mg. A variety of hydrates are known with the formula MgCl2(H2O)x, and each loses water with increasing temperature: x = 12 (−16.4 °C), 8 (−3.4 °C), 6 (116.7 °C), 4 (181 °C), 2 (ca. 300 °C). In the hexahydrate, the Mg2+ remains octahedral, but is coordinated to six water ligands. The thermal dehydration of the hydrates MgCl2(H2O)x (x = 6, 12) does not occur straightforwardly.
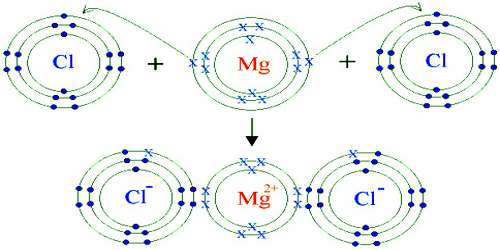
Magnesium Chloride is a colorless crystalline compound. The salt is very hygroscopic. It has density of 2.325 g/cm3 (anhydrous), 1.56 g/cm3 (hexahydrate) and a high melting point of 987 K. Magnesium chloride hexahydrate is only stable below 373 K and decomposes at 391 K. The compound well dissolves in water with solubility of 35.5g per 100g H2O at 298 K. Dissolving MgCl2 is an exothermic process.
Magnesium chloride is very corrosive to the metal surfaces of automobiles and causes rust if left on the vehicle for a long period of time. It is thicker than sodium chloride, so it sticks to a car’s finish and is almost impossible to wash off.
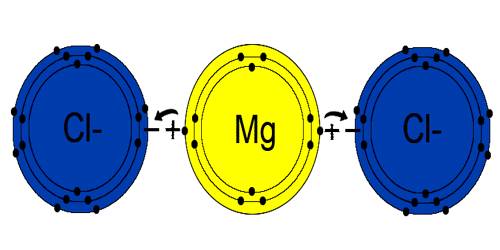
Applications of Magnesium Chloride
Magnesium chloride is mostly used for dust control, ice control and road stabilization. It is also used to produce magnesium metal, fertilizer, artificial seawater, feed supplements, textiles, paper, fireproofing agents, cements and refrigeration brine. It is also used for wastewater treatment.
Magnesium cations Mg2+ play an important role in many cellular functions particularly as cofactors in enzymatic reactions. Magnesium is essential for normal cardiovascular and muscular activity. Doctors prescribe magnesium salts as prophylactic and clinical treatment.

The application of a magnesium chloride solution to external wounds had favorable effects over leucocytic activity and phagocytosis, making it ideal for wound cleansing. Oral magnesium chloride was a powerful immuno-stimulant, exhibiting a broad tonic effect on the host.
Reference: