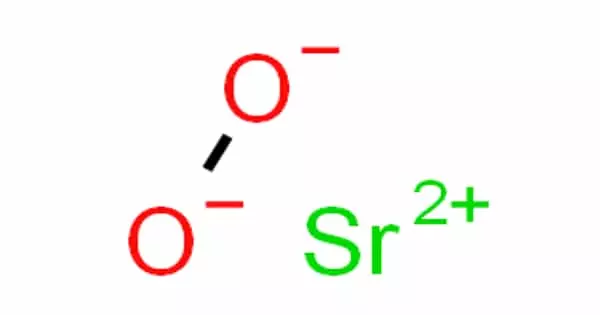
Strontium peroxide is an inorganic compound with the formula SrO2 that exists as an anhydrous and octahydrate white solid. The structure of the anhydrous form is similar to that of calcium carbide. When heated and in contact with organic substances, it can cause fires or explode. It functions as both a reducing and an oxidizing agent.
Strontium peroxide appears in the form of a white powder. It is insoluble in water and decomposes slowly in water. Noncombustible, but hastens the combustion of combustible material.
Properties
Strontium peroxide is a light-colored solid of good thermal stability. Commercial strontium peroxide contains about 85% SrO2 and 10% active oxygen. It is insoluble in water and slowly decomposed by water.
- Compound Formula: O2Sr
- Molecular Weight: 119.62
- Appearance: White Powder
- Melting Point: 215 °C
- Boiling Point: N/A
- Form: Powder
- Color: Pale brown
- Density: 4.56 g/mL at 25 °C
- Solubility in H2O: N/A
- Monoisotopic Mass: 119.895447
Production
Strontium peroxide is produced by passing oxygen overheated strontium oxide. Upon heating in the absence of O2, it degrades to SrO and O2. It is more thermally labile than BaO2.

Preparation
Like the other alkaline earth peroxides, it can be prepared by reaction of the nitrate and sodium peroxide in a cold solution:
Sr(NO3)2 + Na2O2 +xH2O
SrO2·xH2O+2NaNO3
The hydrated form is usually the octahydrate. If the anhydrate is desired, the hydrated peroxide is dried and then sintered at 350°C for 10 min or less:
SrO2·xH2O + heat → SrO +SrO2 +H2O
Uses
Strontium Peroxide is used in pyrotechnics, such as fireworks (red color), ammunition (tracer/hunting), oxidizers, and alarm rockets. It’s also used in textiles to bleach fibers and as an oxidizing agent in hair bleaching. This compound’s only significant application is in pyrotechnics. In flames, strontium peroxide produces a red color.
It is an oxidizing agent that is used in bleaching. It is used as an oxidizer and a bright red pyrotechnic colorant in some pyrotechnic compositions. It’s also useful as an antiseptic and in tracer munitions.
Health Hazard
Friction, heat, or contamination can cause strontium peroxide to explode. When involved in a fire, it hastens the burning process. It may cause combustibles to ignite (wood, paper, oil, clothing, etc.). The mixture may be explosive if the combustible material is finely divided.
It is toxic; inhaling, ingesting, or coming into contact (skin, eyes) with the vapors, dust, or substance can result in severe injury, burns, or death. Fire can emit irritants and/or toxic gases. Toxic fumes or dust can build up in confined spaces (basements, tanks, hopper/tank cars, and so on). Pollution may occur as a result of runoff from firefighting or dilution of water.