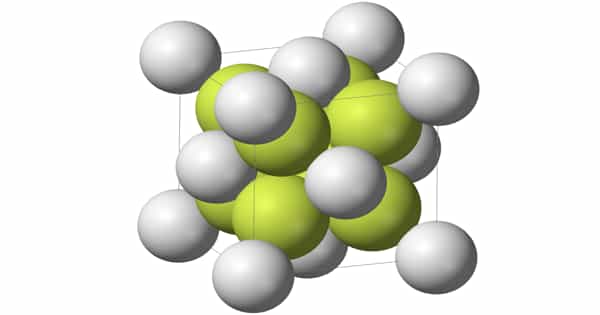
Strontium fluoride, SrF2, is a strontium fluoride that is also known as strontium difluoride and strontium(II) fluoride. It is a white crystalline solid that is brittle. It can be found in nature as an extremely rare mineral strontiofluorite. It has a broad range of transparency, from ultraviolet to long-wave infrared, with low reflectance loss and dispersion.
Properties
It is almost completely insoluble in water (its Ksp value is approximately 2.0×10-25 at 25 degrees Celsius). It irritates the eyes and skin and is toxic if inhaled or ingested. SrF2, like CaF2 and BaF2, exhibits superionic conductivity at high temperatures.
Strontium fluoride is light transparent at wavelengths ranging from vacuum ultraviolet (150 nm) to infrared (11 µm). It has optical properties in between calcium fluoride and barium fluoride.
- Compound Formula: F2Sr
- Molecular Weight: 125.62
- Appearance: White
- Melting Point: 1,477° C (2,691° F)
- Boiling Point: 2,460° C (4,460° F)
- Density: 4.24 g/cm3
- Solubility in H2O: N/A
- Exact Mass: 125.902 g/mol

Preparation
Hydrofluoric acid reacts with strontium carbonate to produce strontium fluoride. It is made by reacting strontium chloride with fluorine gas or by acting on strontium carbonate with hydrofluoric acid. It irritates the eyes and skin and is toxic if inhaled or ingested.
Structure
The fluorite structure is used in the solid. The SrF2 molecule is nonlinear in the vapor phase, with an FSrF angle of about 120°. This deviates from VSEPR theory, which predicts a linear structure. According to ab initio calculations, contributions from d orbitals in the shell below the valence shell are to blame. Another theory is that polarization of the strontium atom’s electron core produces an approximately tetrahedral charge distribution that interacts with the SrF bonds.
Uses
Strontium fluoride is used as an optical material in a limited number of applications, such as as an optical coating on lenses and as a thermoluminescent dosimeter crystal.
It is used in a few special applications as an optical material that is transparent from vacuum ultraviolet (150 nm) to infrared (11 µm). It has optical properties in between calcium fluoride and barium fluoride. It is also used on lenses as an optical coating.
Strontium fluoride is used as a carrier of the radioisotope strontium-90 in radioisotope thermoelectric generators. Another application is as a carrier of the radioisotope strontium-90 in radioisotope thermoelectric generators.