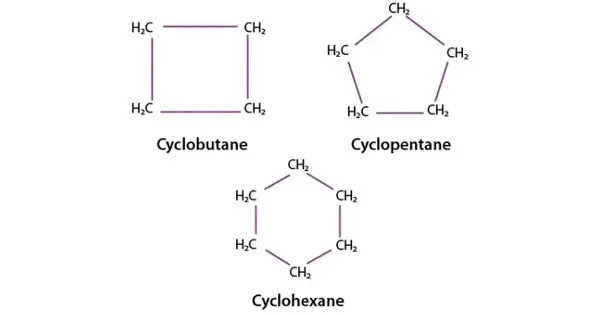
A cycloalkyne is a cyclic counterpart of an alkyne (−C≡C−). A cycloalkyne is a closed ring of carbon atoms with one or more triple bonds. These are cyclic hydrocarbons with at least one triple bond (alkyne functional group) within their ring structures. They form a subset of cyclic alkynes. Cycloalkynes, like other alkynes, have triple-bonded carbon atoms that are sp hybridized.
Cycloalkynes have the generic formula CnH2n-4. Cycloalkynes are highly strained due to the linear structure of the C−C≡C−C alkyne unit. They can only exist when the amount of carbon atoms in the ring is sufficient to accommodate this geometry. Large alkyne-containing carbocycles may be virtually unstrained, yet the tiniest members of this type of molecules may be subjected to such strain that experimental observation is impossible.
Cyclooctyne (C8H12) is the smallest cycloalkyne capable of being isolated and stored as a stable compound. Despite this, smaller cycloalkynes can be produced and trapped through reactions with other organic molecules or through complexation to transition metals.
The general formula for cycloalkynes is CnH2n-4, where n represents the number of carbon atoms in the ring. For example:
- Cyclooctyne: C8H10 (the smallest cycloalkyne)
- Cyclodecyne: C10H12
- Cyclododecyne: C12H14
Cycloalkynes are relatively rare compared to their acyclic counterparts due to the strain imposed by both the cyclic structure and the presence of the triple bond. The high strain in these molecules makes them highly reactive and potentially explosive. Consequently, they are often studied for their synthetic potential and unique reactivity in organic chemistry.
Cycloalkynes can have unusual reactivity due to ring strain and the presence of a triple bond. They can undergo addition reactions analogous to alkenes, as well as standard alkyne reactions including hydrogenation, hydration, and halogenation. Because of their stretched structure, cycloalkynes are frequently difficult to synthesize and may have distinct chemical characteristics when compared to their acyclic cousins. Their different reactivity patterns make them interesting in organic synthesis and chemical research.