A breakthrough in understanding the sugar biology of multicellular organisms would refer to significant new insights or discoveries in the field of glycobiology, which is the study of carbohydrates and their role in biology.
In multicellular organisms, there are three types of protein glycosylation. N-glycosylation, O-mannosylation and C-mannosylation. In each of these activities, which all take place in the endoplasmic reticulum, enzymes link sugar residues to particular locations on newly formed proteins.
While N- and O-glycosylation are well studied, the third form, C-mannosylation of tryptophan side chains, has long been a mystery to researchers. Although membrane proteins and 20% of all secretory proteins are impacted, the purpose of the shift, how the distinct protein sequences are identified, and how the accompanying enzyme reaction is even chemically conceivable have all remained unknown until recently.
In an international collaboration, researchers from ETH Zurich, the Walter and Eliza Hall Institute of Medical Research (WEHI) in Australia, the University of Chicago and the University of Bern have now elucidated the structure and function of the responsible enzyme, ‘tryptophan C-mannosyltransferase’ (CMT). The corresponding study was published in the latest issue of the journal Nature Chemical Biology.
CMT is a member of the category C (GT-C) glycosyltransferase enzymes, one of the three glycosyltransferase superfamilies. The most prominent member is oligosaccharyltransferase (OST), which is responsible for N-glycosylation.
The CMT identifies highly specific protein sequences similarly to the OST, with the exception that in mammals, four distinct CMTs coexist simultaneously and recognize various protein sequences.
Sugars help immuno-receptors to the cell surface
The requisite equipment, including specialized antibodies and mass spectrometry test procedures, have just recently been created in order to be able to determine the degree of C-mannosylation. It was demonstrated that this mechanism virtually exclusively takes place in areas where cell-cell contact is crucial, particularly in adhesion GPCRs and immune system cytokine receptors. The latter serve as “sensory antennae” for growing neurons that make their way through the brain.
“The topic is red-hot, especially for our understanding of the cell-cell communication of the immune system,” explains Kaspar Locher, Professor of Structural Biology at ETH Zurich: “Signaling molecules such as cytokines direct the immune response during an infection. While these and their associated receptors have been intensively studied for decades, it has long been neglected that C-mannosylation determines whether a cytokine receptor reaches the cell surface to exert its function.”
“With our insights into the structure of the enzymes involved, we now have a near-complete understanding of how C-mannosylation gets to these receptors,” adds study first author Joël Bloch, former senior scientist in Locher’s group.
Tailor-made molecular construction kit
The ETH researchers succeeded in producing the CMT enzyme in its pure form. Chemists from WEHI (AUS) and the University of Bern worked with them to create unique compounds that imitate sugar substrates and protein sequences that are peculiar to CMT. This gave them the chance to examine the enzyme’s unique characteristics for the first time in a test tube.
The researchers quickly realized that the enzyme chemistry of CMT must be novel and completely different from that of OST.
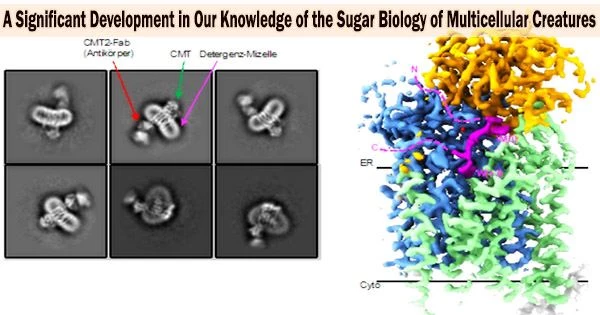
“In such a case, we can only find out the mechanism of an enzyme using high-resolution structural elucidation. The problem, however, was that CMT could not be crystallized until now and had too little mass for cryo-EM, because this technique is particularly difficult to apply to proteins below 100 kDa,” Locher explains.
Antibody enables high-resolution electron microscopy
A trick finally brought the breakthrough: The ETH scientists created a synthetic antibody that attaches precisely to the CMT in association with colleagues from the University of Chicago. The enzyme’s mass was boosted by this antibody to the point that cryo-EM could be used to determine the enzyme’s structure. The team led by Kaspar Locher was able to ultimately understand how the various CMT variations detect various protein sequences with the use of the cryo-EM structures.
These new understandings allowed the researchers to more accurately predict which human proteins had the change. From this, they hope to be able to capture the ‘C-mannosyl proteome’ in the near future.
The researchers want to advance the development of CMT-specific inhibitors by unraveling the peptide binding mechanism of CMTs. Such molecules could aid in the development of drugs to treat diseases like malaria, where Plasmodium falciparum has its own CMT and depends on it to attach to the host.
Given that it is essential for the production of sperm, the sequence and organ specificity of the human CMT variation CMT2 could also be utilized. Therefore, CMT2 inhibitors could be developed as male contraceptives using the latest discoveries.
A novel enzyme mechanism
The enzymatic mechanism of CMT presented scientists with yet another puzzle. This results in a distinctive carbon-carbon link between sugar and protein. The researchers were able to “capture” CMT in the midst of a glycosyl transfer reaction and reveal its cryo-EM structure by using a specially produced CMT inhibitor molecule.
They were then able to see how the CMT reaction works a previously unidentified type of electrophilic aromatic substitution made possible by well placed side chains. These discoveries may aid in the creation of custom enzymes that catalyze bonding between carbon atoms.
Evolutionarily conserved protective mechanism in glycosyltransferases
With a total of four different structures of the CMT, the scientists succeeded for the first time in visualizing a practically complete catalytic cycle of an enzyme of the GT-C superfamily.
In the process, they uncovered an astonishing mechanism: Due to their lipid binding, the sugar substrates of the CMT are difficult to manufacture and consequently of particular value. It turned out that the CMT first binds them in a shielded, non-reactive binding pocket.
The sugar substrate is moved by a peptide sensor and delivered into a highly reactive condition only when the protein or peptide to be modified docks onto the CMT.
The researchers believe that this mechanism, which protects priceless substrate molecules from being used too soon, is evolutionarily conserved in GT-C enzymes.
“Having uncovered the general architecture of GT-C enzymes three years ago, we now have a holistic understanding of their enzyme chemistry. It is another milestone in glycobiology,” explains Locher.