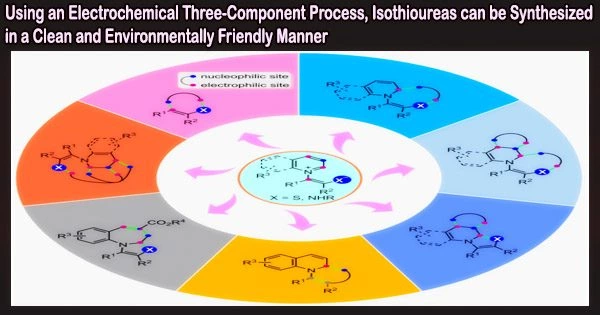
Isothiourea molecules serve as a crucial synthesis intermediate in both agriculture and medicine. Isothioureas can function as ligands or organocatalysts in transition metal catalysis in addition to being strong inhibitors of biological targets.
A team of Chinese researchers describes a novel electrocatalytic three-component reaction approach for the synthesis of isothioureas in a study that was just published in the journal Green Synthesis and Catalysis.
In comparison to traditional procedures, multicomponent reactions provide advantages such as operational simplicity, cost-effective steps, avoiding laborious purification processes, energy efficiency, and minimal waste formation.
The use of heavy metal catalysts or stoichiometric oxidants as processes’ main oxidants limits the sources of sulfur among the published methods for the synthesis of isothioureas via multi-component reactions.
Using thiols, isocyanides, and amines as substrates, the team led by Professors Yingming Pan and Haitao Tang from the School of Chemistry and Pharmaceutical Sciences at Guangxi Normal University discovered that under electrochemical conditions, isothiourea derivatives can be synthesized through free radicals.
When isocyanides were attacked by radicals, they formed imine carbon radical active intermediates, and reacted with aniline to synthesize isothioureas. The synthesis strategy of more complex isothioureas was constructed by controlling the formation of iminocarbon radical active intermediates from isocyanides by the electrochemical method.
Professors Yingming Pan
Notably, this reaction method has the benefits of broad substrate applicability, robust functional group tolerance, and great atomic economy and does not require heavy metal catalysis or stoichiometric oxidants.
With this novel method, isothioureas were produced by electrochemically oxidizing thiophenols with varied substituents to sulfur radicals while using isocyanides with diverse reactive sites as substrates.
“When isocyanides were attacked by radicals, they formed imine carbon radical active intermediates, and reacted with aniline to synthesize isothioureas,” explained Pan. “The synthesis strategy of more complex isothioureas was constructed by controlling the formation of iminocarbon radical active intermediates from isocyanides by the electrochemical method.”
According to Muxue He, a researcher involved in this study, this is a major breakthrough in the construction of isothiourea derivatives through multi-component reactions. “So far, multicomponent construction of isothioureas has required the use of heavy metal catalysts and stoichiometric oxidants. Our method shows that these reagents can be avoided by means of electrochemical synthesis.”
“We hope that our results will encourage scientists to continue to investigate facile and efficient synthetic strategies for isothioureas,” added Tang.