Sodium Hypochlorite
Definition
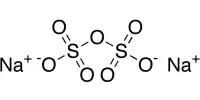
Sodium Hypochlorite (NaOCl·5H2O) is an unstable salt that is usually stored in solution and used as a fungicide and oxidizing bleach. It is a colorless or slightly yellow watery liquid with an odor of household bleach. It is produced by a continuous process will have approximately 0.2% by weight excess sodium hydroxide, resulting in a specific gravity of 1.160 at 120 gpl. It is practically and chemically distinct from chlorine. Sodium hypochlorite is frequently used as a disinfectant or a bleaching agent.
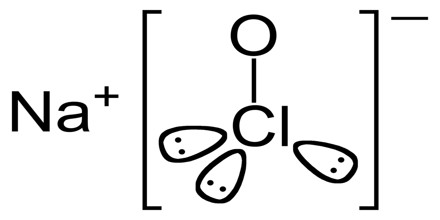
Sodium Hypochlorite can be produced using either a batch production method, such as with the Powell Sodium Hypochlorite Batch System or in greater volumes using a continuous method. Continuous production of sodium hypochlorite can be achieved with the Powell Continuous Sodium Hypochlorite Dry Liquid/Vapor System or with the Powell Continuous Sodium Hypochlorite System that uses wet vapor chlorine from electrolytic cells. It can be easily produced for research purposes by reacting ozone with salt.
NaCl + O3 → NaClO + O2
This reaction happens at room temperature and can be helpful for oxidizing alcohols.
Sodium Hypochlorite (NaClO) is produced by a process known as the Hooker process, which is the method used for a large-scale production of NaClO. In the Hooker process, sodium hydroxide (NaOH) is exposed to chlorine gas (Cl2). Along with sodium hypochlorite, the reaction between these two substances produces sodium chloride (NaCl) and water (H2O).

Uses and Safety of Sidium Hypochlorite
The most common application for sodium hypochlorite is its use as the active ingredient in bleach. The majority of the sodium hypochlorite that is produced is for use as household bleach, which commonly contains 5% sodium hypochlorite. It is used extensively as a bleaching agent in the textile, detergents, and paper and pulp industries. It is also used as an oxidizing agent for organic products. In the petrochemical industry, sodium hypochlorite is used in petroleum products refining. Large quantities are also used as a disinfectant in water and wastewater treatment and sanitary equipment. In food processing, sodium hypochlorite is used to sanitize food preparation equipment, in fruit and vegetable processing, mushroom production, hog, beef and poultry production, maple syrup production, and fish processing.

Sodium Hypochlorite is a strong oxidizer. Oxidation reactions are corrosive. Solutions burn the skin and cause eye damage, especially when used in concentrated forms. However, as recognized by the NFPA, only solutions containing more than 40% sodium hypochlorite by weight are considered hazardous oxidizers. Solutions less than 40% are classified as a moderate oxidizing hazard.
Reference: study.com, dictionary.com, powellfab.com, wikipedia.