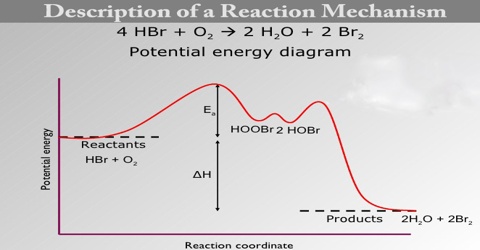
Description of a Reaction Mechanism
A reaction mechanism describes the one or more steps involved in the reaction in a way which makes it clear exactly how the various bonds are broken and made. The following example comes from organic chemistry.
In any chemical change, some bonds are broken and new ones are made. Quite often, these changes are too complicated to happen in one simple stage. Instead, the reaction may involve a series of small changes one after the other.
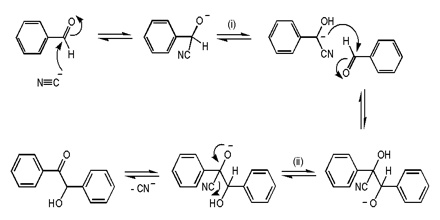
This is a reaction between 2-bromo-2-methylpropane and the hydroxide ions from sodium hydroxide solution:
![]()
The overall reaction replaces the bromine atom in the organic compound by an OH group.
The first thing that happens is that the carbon-bromine bond in a small proportion of the organic compound breaks to give ions:
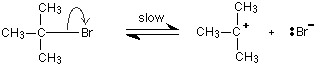
Carbon-bromine bonds are reasonably strong, so this is a slow change. If the ions hit each other again, the covalent bond will reform. The curly arrow in the equation shows the movement of a pair of electrons.
If there is a high concentration of hydroxide ions present, the positive ion stands a high chance of hitting one of those. This step of the overall reaction will be very fast. A new covalent bond is made between the carbon and the oxygen, using one of the lone pairs on the oxygen atom.
A mechanism must satisfy the following two requirements:
- The elementary steps must add up to give the overall balanced equation for the reaction.
- The rate law for the rate-determining step must agree with the experimentally determined rate law.
Each of these events constitutes an elementary step that can be represented as a coming-together of discrete particles (“collision”) or as the breaking-up of a molecule (“dissociation”) into simpler units. The molecular entity that emerges from each step may be a final product of the reaction, or it might be an intermediate.
Key Points of Reaction Mechanism
The reaction mechanism describes the sequence of elementary reactions that must occur to go from reactants to products.
Reaction intermediates are formed in one step and then consumed in a later step of the reaction mechanism.
The slowest step in the mechanism is called the rate determining or rate-limiting step.
The overall reaction rate is determined by the rates of the steps up to (and including) the rate-determining step.
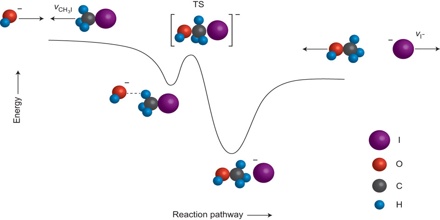
Relating Reaction Mechanisms to Rate Laws
It’s often the case that one step in a multistep reaction mechanism is significantly slower than the others. Because a reaction cannot proceed faster than its slowest step, this step will limit the rate at which the overall reaction occurs. The reaction mechanism describes the sequence of elementary steps that occur to go from reactants to products. Let’s start by considering the following reaction between nitrogen dioxide and carbon monoxide:
NO2(g) + CO(g) → NO(g) + CO2(g)
Based on the balanced reaction, we might hypothesize this reaction might occur from a single collision between a molecule of nitrogen dioxide and a molecule of carbon monoxide. In other words, we hypothesize this elementary reaction.
Reaction Mechanisms and the Rate Law
A complex reaction is a little like building a car on an assembly line, where each assembly step is a molecular collision. Each collision can result in the breaking and/or making of one or more chemical bonds.
Chemists can come up with a hypothetical reaction mechanism based on the experimentally determined rate law, as well as chemical intuition. At a minimum, the elementary reactions that make up the proposed reaction mechanism must sum to the overall reaction.
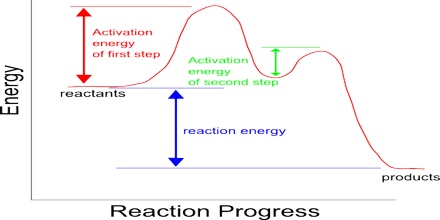
Summary
The reaction mechanism describes the sequence of elementary reactions that must occur to go from reactants to products.
Reaction intermediates are formed in one step and then consumed in a later step of the reaction mechanism.
The slowest step in the mechanism is called the rate determining step or rate-limiting step.
The overall reaction rate is determined by the rates of the steps up to (and including) the rate-determining step.