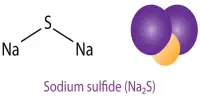
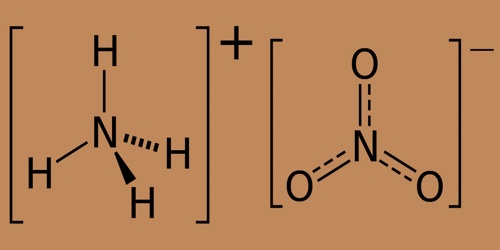
Ammonium nitrate is the nitric acid ammonium salt; it is a chemical compound containing the chemical formula NH4NO3. At room temperature, it is a colorless rhombic or monoclinical crystal. It can be degraded at 210°C to water and nitrous oxide. They are prone to decomposition into nitrogen, oxygen, and water after intense heating at 300°C above. Predominantly used as a high-nitrogen fertilizer in agriculture. In 2017 global production was estimated at 21.6 million tonnes.
Ammonium nitrate plays a role as a fertilizer, an explosive agent, and an oxidizer. It is an inorganic molecular form, salt with ammonium, and salt with inorganic nitrate. The other main use is as an explosive component of mixtures used in mining, quarrying, and civil construction. It is the main constituent of ANFO, a preferred industrial explosive which accounts for 80% of explosives employed in North America. It’s soluble in water, methanol, and ethanol. It dissolution in water can absorb plenty of warmth and reduce the temperature. It’s one in all the foremost nitrogen fertilizer varieties within the world today.
Ammonium nitrate is present as the natural mineral gwihabaite, the saltpetre ammonium analog in the driest regions of the Chilean Atacama Desert, sometimes as a crust on the ground or in combination with other minerals of nitrate, iodate, and halides. It is commercially available both as a colorless crystalline solid and for particular applications is transformed into prills. Ammonium nitrate was mined there within the past, but virtually 100% of the chemical now used is synthetic. Nitrate has no residue within the soil, and maybe all absorbed by crops; being a physiological neutral fertilizer.
The ammonium nitrate is used in protection bombs, matches, and pyrotechnics as a pesticide, as a freezing mixture. It is suitable for a wide variety of soils and crops but is best suited for dry and dry crops especially suited for cash crops such as tobacco, cotton, and vegetables. The industrial production of ammonium nitrate entails the acid-base reaction of ammonia with nitric acid:
HNO3 + NH3 → NH4NO3
Ammonium nitrate should not be used as basal fertilizer, since the dissociated nitrate ions are quickly leached into the soil after being added to the soil. It is used in its anhydrous (a gas) form and concentrated nitric acid. Due to its highly exothermic nature, the reaction is aggressive. Ammonium nitrate is dissolved in water, the detonation sensitivity is greatly reduced, for which it is impossible to detonate the ammonium nitrate with human power.
As ammonium nitrate is a salt, both the cation, NH4+ and the anion, NO3−, may take part in chemical reactions. Ammonium nitrate is a highly reactive and durable high explosive that is used as a slow-burning rocket propellant when combined with burning rate catalysts. But it is also used in insecticides, pyrotechnics, rust inhibitors, and. The ammonium nitrate melts at 337 ° F (170 ° C) and starts to decompose when molten. Hazardous ammonium nitrate scenarios may include simple thermal decomposition caused by external fire or other heating, self-sustaining decomposition also known as “cigar burning”, and detonation.
Most types of ammonium nitrate don’t still decompose once a fireplace has been extinguished. Nitrate features a critical ratio of 59.4%, above which it’ll absorb moisture from the atmosphere. Therefore, it’s important to store nitrate in a very tightly sealed container. However, some forms of ammonium nitrate fertilizers containing a small percentage of chlorides (e.g., potassium chloride) undergo a smoldering (self-sustaining) decomposition that can spread across the mass to create significant toxic fumes, even when the initial heat source is removed.
Information Sources: