Researchers at UT Southwestern have discovered a four-protein complex that appears to play an important function in the formation of ribosomes (organelles that serve as protein factories for cells) as well as a surprise role in neurodevelopmental diseases. These results, which were reported in Cell Reports, could lead to novel strategies to manipulate ribosome production, which could have an impact on a range of human health disorders.
“Ribosomes are essential for life, but we’ve had an incomplete understanding of how they’re assembled and how the process of ribosome production is regulated,” said lead author Michael Buszczak, Ph.D., Professor of Molecular Biology and member of UT Southwestern’s Harold C. Simmons Comprehensive Cancer Center. “Our findings shed a lot of light on these issues.”
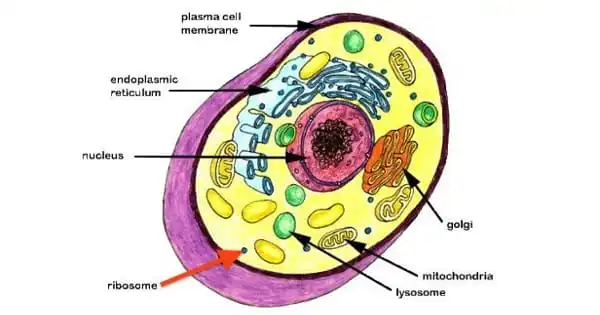
A ribosome is composed of RNA and proteins, and each ribosome is made up of two distinct RNA-protein complexes known as the small and big subunits. The large subunit is stacked on top of the small subunit, with an RNA template sandwiched between them.
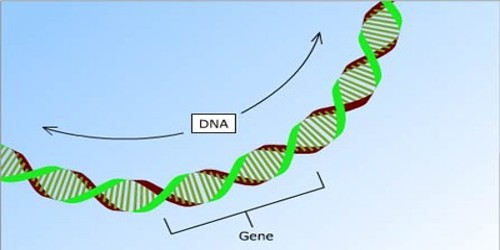
Ribosomes in eukaryotes get protein synthesis orders from the nucleus, where parts of DNA (genes) are transcribed to produce messenger RNAs (mRNAs). An mRNA is transported to the ribosome, which uses the information contained inside it to construct a protein with a specific amino acid sequence. This is referred to as translation. Because prokaryotes lack a nucleus, their mRNAs are produced in the cytoplasm and are instantly translated by ribosomes.
Ribosomes are essential for life, but we’ve had an incomplete understanding of how they’re assembled and how the process of ribosome production is regulated. Our findings shed a lot of light on these issues.
Professor Michael Buszczak
Ribosomes are found in variable numbers in every cell of every life on Earth, according to Dr. Buszczak. Variations from these natural set points can have negative implications because of their critical role as protein makers, he warned. Cancer cells, for example, tend to increase ribosome production in order to enhance protein production, which is required for unregulated cell proliferation. Furthermore, ribosomopathies, a group of rare disorders characterized by aberrant ribosome production, manifest with a variety of symptoms such as anemia, craniofacial deformities, and intellectual incapacity.
Although ribosomes exist in many species, the majority of what is known about ribosome biogenesis has come from the common lab model, yeast. The fundamentals of this process are the same for human ribosome biogenesis, according to Dr. Buszczak, but the specifics are not. As a result, the characteristics that distinguish human ribosome production have remained unknown.
Dr. Buszczak, Chunyang Ni, a graduate student in the Buszczak lab, and their colleagues, including Jun Wu, Ph.D., Assistant Professor of Molecular Biology at UTSW, began by developing a technique that caused old ribosomes to glow red and newly generated ribosomes to glow green to learn more about this process. The researchers employed this method on a variety of human cell types, confirming that the rates of ribosome manufacturing differed between them.
The researchers used the CRISPR gene-editing technology to inactivate specific genes in order to identify those that could be important participants in ribosome biogenesis. Their search yielded four genes: CINP, SPATA5L1, C1orf109, and SPATA5. Further investigation revealed that these genes form a complex that removes a placeholder protein from ribosomes when the assembly is nearly complete, allowing a new protein to take its place during ribosome maturation.
Ribosomes are found in almost every cell type of multicellular creatures, as well as prokaryotes such as bacteria because protein synthesis is a fundamental function of all cells. However, eukaryotic cells that specialize in protein production have a disproportionately large number of ribosomes. Because the pancreas is in charge of manufacturing and secreting vast amounts of digestive enzymes, the pancreatic cells that produce these enzymes contain an extremely high number of ribosomes.
The role of SPATA5 in cells was previously unknown; nevertheless, mutations in this gene have been linked to neurodevelopmental diseases such as microcephaly, hearing loss, epilepsy, and intellectual disability. When the researchers injected two of these mutations into cells, leading them to produce a mutant SPATA5 protein, the cells were unable to synthesize the typical quantity of functioning ribosomes, implying that these neurodevelopmental diseases may be caused by ribosome issues.
Dr. Buszczak and his colleagues intend to investigate why the central nervous system appears to be more sensitive to ribosomal disturbances than other cell types. He went on to say that these findings could potentially lead to new treatments for cancer, ribosomopathies, and other diseases caused by protein over- or under-production.