To create a new drug at breakneck speed, a combination of advanced techniques, efficient processes, and medicinal chemistry expertise is required. Researchers use rapid biphasic (gas and liquid) mixing to create a fluorine-based compound.
How long is a second? A second is defined as one 86,400th of a 24-hour day. A bullet train traveling at 300 kilometers per hour can cover 83 meters in one second. An individual’s blink lasts 0.3 seconds on average, allowing three blinks to occur within one second. A POSTECH-led research team has proposed a method for synthesizing fluorine-based compounds that involve a rapid mixing reaction between a gaseous component and a liquid that takes less than a second.
Professor Dong-Pyo Kim and Jeong-Un Joo (Department of Chemical Engineering at POSTECH) and Professor Heejin Kim and Hyune-Jea Lee (currently a researcher at Samsung Advanced Institute of Technology) from Korea University’s Department of Chemistry have successfully developed a new method for synthesizing trifluoromethyl intermediate (-CF3) from fluoroform (CHF3). It entails the use of a special reactor capable of ultra-fast mixing of gas and liquid. This method holds great promise for the development of new fluorine-based drugs. The findings were published in Nature Communications.
The findings of the study have important implications for industrial applications in the economically efficient synthesis of fluoride compounds, making them more practical and contributing significantly to research on several unstable intermediates.
Fluorine does not exist in its pure form in nature, but only in the form of various chemical compounds. Because of its ability to coat teeth and prevent cavities, sodium fluoride, a fluorine-containing compound, is used as an ingredient in toothpaste. Recent research has highlighted the potential of synthetic drug molecules containing fluorine due to their high permeability into diseased tissue cell membranes and strong binding affinity against proteins. As a result, there is growing interest in the development of fluorine-containing drugs.
There are several methods for producing trifluoromethyl, but the most cost-effective one involves replacing a hydrogen atom from fluoroform, a simple precursor, with another element or functional group. However, because gaseous fluoroform is volatile, it is difficult to mix with liquids and has a low reactivity. Furthermore, it decomposes instantly, necessitating the addition of a substance capable of reacting with it. Unfortunately, this process can cause unintended chemical reactions, resulting in a low trifluoromethyl yield.
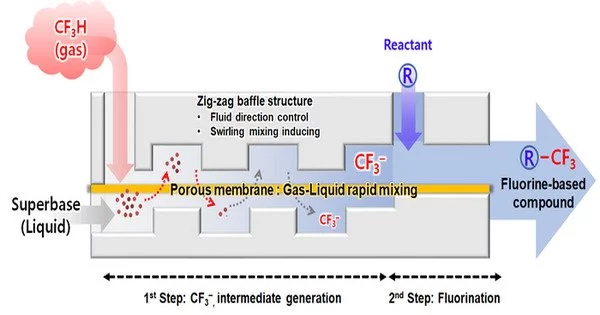
To overcome the difficulty of producing trifluoromethyl from fluoroform, the researchers devised a novel gas-liquid reactor with a zigzag-shaped channel and highly permeable non-porous membranes sandwiched between the upper and lower channels. Within the reactor, this configuration allowed for the swirling and mixing of superbase, a liquid used for dehydrogenation, and gaseous fluoroform.
The team was able to effectively produce trifluoromethyl anion (CF3-) by breaking fluoroform bubbles into smaller pieces to increase the contact area between gas and liquid. Unlike traditional methods, they effectively produced a fluoride intermediate without the use of stabilizers or additives.
The researchers created a fluorine-based compound by adding a compound that will react with the fluoride anion intermediate right away. The entire process of generating a fluorine anion intermediate from fluoroform took less than a second. The researchers increased the formation of a trifluoromethyl anion, which is known to be unstable, and accelerated the subsequent reaction before the intermediate decomposed. This method increased the yield of fluoride-based compounds and introduced a reliable method for the synthesis of fluorine-based drugs.
The findings of the study have important implications for industrial applications in the economically efficient synthesis of fluoride compounds, making them more practical and contributing significantly to research on several unstable intermediates.