If you own a retro gaming console or have an old roll of packing tape, you’ve seen how plastics turn yellow as they age. Though the cause of this color change has long been attributed to the formation of molecules that act as dyes – the actual chemical changes that take place remained unexplained. Now, researchers reporting in ACS Applied Polymer Materials have identified surface-based chiral nanostructures as the potential culprit.
Understanding how and why polymers degrade with age is key to designing alternatives that can avoid these pathways, allowing plastic products to have a longer lifespan. For one of the most commonly used plastics, polyethylene, it’s long been suggested that ultraviolet (UV) light – the same light that gives us sunburns – initiates reactions in the backbone of the polymer’s structure that cause the yellow color change.
The researchers first investigated if potential structures formed on yellowed polyethylene films’ surfaces interacted with circularly polarized light, a type of light whose waves travel with a right- or left-handed rotation.
However, though chemical changes to polyethylene’s polymeric backbone have been observed after exposure to UV light, those new structures cannot account for polyethylene’s yellowing. One emerging way to intentionally modify the color and the ways that plastics interact with light is to create nano-sized “supramolecular” structures on their surfaces that impact plastics’ properties in a controllable way. Inspired by these surface-based technologies, Margaret M. Elmer-Dixon, Melissa A. Maurer-Jones and colleagues wanted to see if such nanostructures formed unintentionally by UV light could be the cause of polyethylene yellowing.
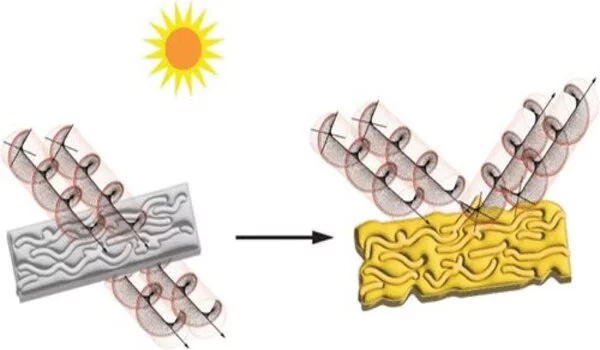
The researchers first investigated if potential structures formed on yellowed polyethylene films’ surfaces interacted with circularly polarized light, a type of light whose waves travel with a right- or left-handed rotation. The amount of circularly polarized light absorbed by the film in these experiments changed depending on the film’s orientation, suggesting that the yellowed plastic contains new chemical structures that are chiral, that is, they are directional and aren’t identical to their mirror images.
Additional experiments showed that most of the degradation during film yellowing occurred on the surface of the films. The team concluded that chiral chemical structures on the surfaces of the polyethylene films are formed during exposure to UV light and are a potential cause for the yellow color of old plastics. They say that these insights could help researchers design plastic products that last longer before becoming unsightly or unusable.
















