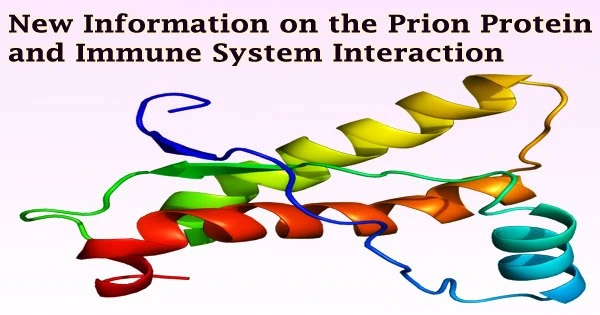
The neurodegenerative disease scrapie can serve as a model for other diseases like Alzheimer’s and Parkinson’s caused by the buildup of proteins that induce tissue malformations (proteinpathies).
Many questions regarding these diseases still remain unanswered.
The development of this disease is linked to a number of factors, including the uptake of the prion protein (PrPSc) and how this protein interacts with immune cells in the intestines, according to a recent doctoral study.
Because it spreads across different animals and results in sponge-like, degenerative alterations in the brain, scrapie in sheep is categorized as a “Transmissible spongiform encephalopathies” (TSE) illness.
Not just sheep, but also cattle (BSE), deer (CWD), and humans (CJD) are affected by these diseases. As was the situation in the 1990s when more than 200 people were exposed to CJD through food, they can also, to a certain extent, be spread between animals.
TSE, often referred to as prion disorders, is believed to be spread by a sick form of the prion protein, which is a normal part of body cells and is particularly prevalent in the brain.
In general, prion illnesses can be inherited, infectious, or sporadic/spontaneous. When the healthy prion protein mutates into the diseased form, which is distinct from the healthy prion proteins by its changed structural makeup, illness results.
This prion protein has a unique shape that makes it challenging for the body’s cells to break down, causing it to accumulate. Since PrPSc is discovered in the digestive system’s lymphatic tissue at an early stage of the illness, it is thought that the gastrointestinal tract is the route of transmission.
Caroline Piercey Kesson, a veterinary scientist, shed new light on early disease-development processes while conducting her dissertation research on the prion protein’s absorption in the intestines.
She disproved past theories by proving, using immunoelectron microscopy, that the disease-causing prion protein is not transferred straight from the intestines to the lymphatic tissue connected to the intestines.
On the contrary, she demonstrated that the protein moved freely or within lymphatic cells outside of the intestines’ organized lymphatic tissue. Dendritic cells are thought to act as “gatekeepers” that control what the body can accept and which immune defense responses it needs to mount in response to invading chemicals.
Therefore, one of Åkesson’s project’s goals was to investigate how dendritic cells and prion protein uptake interact. In order to determine which cell types were linked to the uptake of the prion protein, it was first required to characterize dendritic cells in healthy sheep intestines.
Her findings showed that it was not dendritic cells, but macrophages, which were mainly responsible for the uptake of the protein.
According to Åkesson’s research, the prion protein utilizes the intestine’s typical physiological absorption pathway for macromolecules, which may have a considerable impact on the body’s immune defense system.
One potential outcome is the stimulation of immuno-tolerance, which would prevent a typical immune response to the prion protein ingested through the intestines.
Future research that can shed light on the movement of immune cells and the body’s processing of the prion protein will be very valuable in understanding not only scrapie but also other neurodegenerative proteinopathies that affect both humans and animals.
Cand.med.vet. Caroline Piercey Åkesson defended her doctoral thesis on 20th December 2011 at The Norwegian School of Veterinary Science. Studies on the absorption of prions and their early interactions with immune cells of the sheep intestine are the subject of the dissertation.