Le Châtelier’s principle is a principle used in Chemistry and Physics to describe how a system at equilibrium reacts to external changes. It can be used to predict the behavior of a system due to changes in pressure, temperature, or concentration. This is very important, particularly in industrial applications, where yields must be accurately predicted and maximized.
Overview
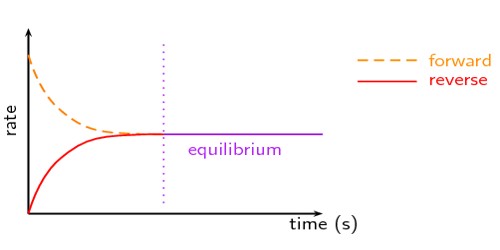
Le Chatelier’s principle is an observation about chemical equilibria of reactions. It is used to show how the point of equilibrium of a system will move to oppose a change in the conditions of the system, following Newton’s third law. This is most commonly seen in reversible reactions in chemistry, where a chemical reaction has reached a point where the formation of products happens at the same rate the products decay back into reactants.
A statement of Le Chatelier’s Principle – If a dynamic equilibrium is disturbed by changing the conditions, the position of equilibrium moves to counteract the change. This principle can then be used to increase the amount of desired products made by moving equilibrium towards that side in chemical industries.
Concentration
According to Le Chatelier’s principle, adding additional reactant to a system will shift the equilibrium to the right, towards the side of the products. If the concentration of reactants in a reaction increases, the point of equilibrium moves towards the products as this gets rid of the excess reactants and cancels the change. Similarly, if the concentration of products increases, the equilibrium moves towards the reactants. Reducing the concentration of any product will also shift the equilibrium to the right.
Pressure
A change in pressure or volume will result in an attempt to restore equilibrium by creating more or fewer moles of gas. If the pressure of a system increases, the point of equilibrium will shift to the side with fewer moles, as that side will occupy less volume and therefore will reduce the pressure. Similarly, if the volume of a system increases or the pressure decreases, the production of additional moles of gas will be favored.
Temperature
The effect of temperature on equilibrium has to do with the heat of reaction. If the temperature of a system is increased, the point of equilibrium moves towards whichever side is more endothermic as this will take in the excess energy as part of the products and so cancels the change of increased temperature. If the temperature of a reaction mixture is changed, the equilibrium will shift to minimize that change.
Electromagnetism
This principle is also shown by Lenz’s law, which states that a changing magnetic flux will produce an opposing magnetic flux (and therefore a magnetic and electric field) in any nearby metals. This will then produce a second current in the nearby metal in the opposite direction to the original current.