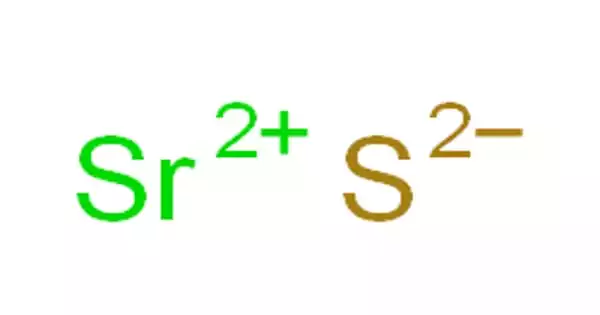Strontium sulfide, abbreviated SrS, is an inorganic compound. It’s a solid white color. It has the odor of rotten eggs. The compound is a step in the process of converting strontium sulfate, the main strontium ore known as celestite (or, more accurately, celestine), to other more useful compounds. It is used to remove hair from the skin and hides as a depilatory.
It reacts violently with acids, producing hydrogen sulfide gas. Toxic gases can be produced when it reacts exothermically with oxidizing agents such as inorganic oxoacids, organic peroxides and epoxides, and inorganic peroxides. There is a moderate risk of fire and explosion.
Properties
Strontium sulfide is a gray powder with a hydrogen sulfide aroma in moist air, slightly soluble in water, soluble in acids (with decomposition), melts above 2000°C, and is used in depilatories, luminous paints, and as a chemical intermediate. Strontium monosulfide is another name for it.
- Melting point: >2000 °C(lit.)
- Density: 3.7 g/cm3
- Form: Powder
- Specific Gravity: 3.7
- Color: Gray
- Water Solubility: Slightly soluble in water. Decomposes in acids
- Sensitive: Hygroscopic

Production and reactions
Strontium sulfide is a grayish-white solid with a thermodynamic Ksp of 500 that reacts with water to form strontium hydrosulfide, Sr(SH)2. Strontium hydrosulfide is formed by (1) reacting strontium sulfide with H2O and (2) saturating strontium hydroxide solution with H2S. Boiling strontium hydrosulfide with sulfur produces strontium polysulfides.
Strontium sulfide is produced by roasting the celestine with coke at 110–1300°C. The sulfate is reduced, leaving the sulfide:
SrSO4 + 2 C → SrS + 2 CO2
About 300,000 tons are processed in this way annually. Both luminous and nonluminous sulfide phases are known, impurities, defects, and dopants being important.
As expected for a sulfide salt of alkaline earth, the sulfide hydrolyzes readily:
SrS + 2 H2O → Sr(OH)2 + H2S
For this reason, samples of SrS have an odor of rotten eggs.
Similar reactions are used to produce commercially useful compounds, such as strontium carbonate, the most useful strontium compound: a mixture of strontium sulfide with either carbon dioxide gas or sodium carbonate results in the formation of strontium carbonate precipitate.
SrS + H2O + CO2 → SrCO3 + H2S
SrS + Na2CO3 → SrCO3 + Na2S
Strontium nitrate can also be prepared in this way.
Uses
In luminous paints; as a depilatory.