Homogeneous catalysts allow greater interaction with the mixture of reactions than heterogeneous catalysts do. Homogeneous catalysis in chemistry is the catalysis by a soluble catalyst in a solution. The advent and rapid growth of new areas of homogeneous catalysis, such as photoredox, electro- and supramolecular catalysis, along with the production of catalysts based on earth-abundant transition metals and elements of the main group.
Catalysts are compounds that, when added to chemical reactions reduce the energy and increase the reaction rate. Homogeneous catalysis refers to reactions where the catalyst is within the same phase because of the reactants, principally in solution. Catalysts lower the energy required to succeed in the transition state of the reaction, allowing more molecular interactions to attain that state. Heterogeneous catalysis, on the other hand, describes processes where the catalysts and the substrates are in distinct phases, usually solid-gas.
In addition to the numerous heterogeneous and enzymatic approaches, homogenous catalysis has emerged as a vital tool for the highly selective transformation of biomass, or biomass-derived platform chemicals. Homogeneous catalysis is a longtime technology that continues to evolve; an illustrative major application is that the production of acetic acid; Enzymes are samples of homogeneous catalysts. However, catalysts don’t affect the degree to which a reaction progresses.
A benefit of homogeneous catalysis is that the catalyst blends into the mixture of reactions, allowing for a very high degree of contact between catalyst and reactant molecules. The proton is a pervasive homogeneous catalyst since the most common solvent is water. By self-ionizing the water, water produces protons. In an illustrative case, acids accelerate (catalyze) the hydrolysis of esters:
CH3CO2CH3 + H2O ⇌ CH3CO2H + CH3OH
At neutral pH, most esters do not hydrolyze the aqueous solutions at acceptable levels. Homogeneous catalysts are used in a number of industrial applications, as they allow for reaction rate increases without temperature increases.
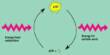
The focus of the organometallic and homogeneous catalysis group is on two overlapping problems – (1) catalysis of industrially large-scale processes like C-H activation of hydrocarbons in petrochemical feedstock (petroleum, natural gas), and (2) catalysis of smaller-scale processes that will result in fine chemicals and advanced organic compounds. Heterogeneous catalysts dominate, but many soluble catalysts are used for stereospecific polymers in particular; homogeneous catalysts are also used in various oxidations. Is the Wacker cycle that produces acetaldehyde from ethylene and oxygen. Recent studies have focused in particular on organometallic complexes of noble metals, such as rhodium, platinum, and gold.
Information Sources: