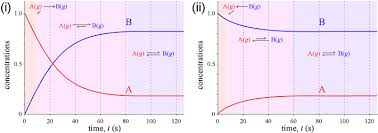
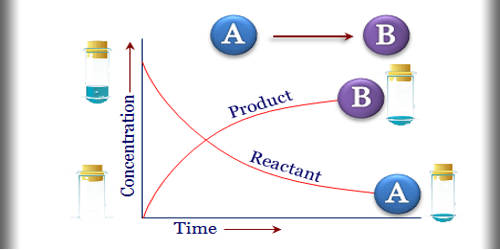
General objective of this article is to Define and Discuss on Equilibrium Concentrations. In a system that has reached chemical equilibrium, the concentrations on the various substances are quantitatively associated. For any equilibrium reaction, the ratio of concentrations on the substances on the right to the concentrations of those around the left equals a constant right for that specific reaction. Observe that the ratio is always written with all the products over the reactants.
Define and Discuss on Equilibrium Concentrations