Blood-type conversion refers to the process of changing one blood type into another. The process that is informed by crystallography is a new development that is currently in pre-clinical trials, meaning it has not yet been tested in humans.
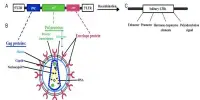
In 2019 Dr. Stephen Withers and colleagues at the University of British Columbia identified a series of enzymes that can be used to modify the chemical structure of a sugar antigen on the surface of blood cells, thereby converting a Type A blood cell to a Type O blood cell the universal donor type. To learn more about how the enzymes produce this alteration, the scientists employed crystallography on the CMCF beamline at the CLS. They then published their findings in Nature Microbiology.
The enzymes Withers and colleagues discovered are extremely effective at making this conversion both on the surface of red blood cells and on the surface of donated human organs such as the lungs or kidneys. These identical antigens are also present on the surface of solid organs.
The company Avivo, which was established to commercialize this technology, is currently engaged optimizing both applications. If successful, this innovative development would mark a significant advancement in the fight against the global and national shortages of blood and organs.
“The idea is that we could broaden the supply considerably,” says Withers, a professor in the Departments of Chemistry and Biochemistry and the Michael Smith Laboratories at UBC. “It would remove the need to worry about blood type in transfusions (and organ donations).”
John Barclay, VP of business development with Avivo says the company is focusing first on applying their approach to organ donations because it’s considerably more straightforward to remove the conversion enzymes prior to transplantation than it is to remove them before transfusing blood.
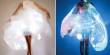
We’ve shown that we can remove those antigens and convert an A type lung to an O type lung quite readily. We’re working on kidneys at the moment…so that’s very exciting.
Dr. Stephen Withers
When a donor organ is harvested, it is frequently mounted on a perfusion device that continually circulates a preservation fluid through it to preserve the viability of the tissue. The Withers team’s enzymes can be added to the fluid combination, where they effectively change the organ’s blood type to the universal blood type.
Following that conversion, the organ is basically “rinsed” out of the solution, which includes the enzymes, as part of the standard transplant procedure. Removing the enzymes from red blood cells or whole blood is considerably more challenging, says Barclay.
The Avivo team has used a set of human lungs that were judged unsuitable for transplant into a patient to show how the process functions. “We’ve shown that we can remove those antigens and convert an A type lung to an O type lung quite readily,” says Withers. “We’re working on kidneys at the moment…so that’s very exciting.”
This application of their technique is in pre-clinical trials now; they’re hoping to move on to clinical trials (i.e., in human patients) in 2024.
“The information we learned from it (crystallography) was very supportive in knowing exactly the structure of the enzymes we’re adding,” says Withers. This information, he says, will be very useful if they need to modify the structure of the enzyme.
It will also be valuable when they seek regulatory approval, to be able to present the complete structure of the enzymes. “We’ve learned a lot more through having that information, which may be useful in the future,” says Withers.
According to Barclay, they are currently conducting study before using the method for blood transfusions. The United States Air Force has given the Withers lab a funding to create an inline filtering device to remove the enzymes from red blood cells or entire blood so that the technology fits inside current blood processing workflows.