Copper sulfate is an inorganic compound that combines sulfur with copper. They are the inorganic compounds with the chemical formula CuSO4(H2O)x, where x can range from 0 to 5. The pentahydrate (x = 5) is the most common form. It is a very versatile chemical with as extensive a range of uses in industry as it has in agriculture. Older names for this compound include blue vitriol, bluestone, the vitriol of copper, and Roman vitriol. It can kill bacteria, algae, roots, plants, snails, and fungi.
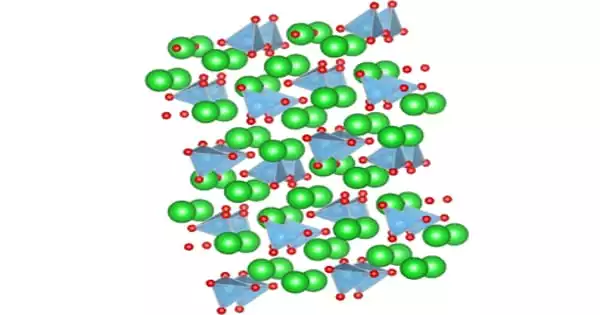
The pentahydrate (CuSO4·5H2O), the most commonly encountered salt, is bright blue. In its solid, crystal-shaped stone form (known as a pentahydrate) it’s known as bluestone or blue vitriol for its blue color. It exothermically dissolves in water to give the aquo complex [Cu(H2O)6]2+, which has octahedral molecular geometry. The structure of the solid pentahydrate reveals a polymeric structure wherein copper is again octahedral but bound to four water ligands. This form is characterized by its bright blue color. However, it can be noted that the anhydrous form of this salt is a powder that is white. The Cu(II)(H2O)4 centers are interconnected by sulfate anions to form chains. Anhydrous copper sulfate is a light grey powder.

Preparation
Copper sulfate is produced industrially by treating copper metal with hot concentrated sulfuric acid or its oxides with dilute sulfuric acid. It can be prepared by treating metallic copper with heated and concentrated sulphuric acid, or by treating the oxides of copper with dilute sulphuric acid. For laboratory use, copper sulfate is usually purchased. Copper sulfate can also be produced by slowly leaching low-grade copper ore in the air; bacteria may be used to hasten the process.
Commercial copper sulfate is usually about 98% pure copper sulfate and may contain traces of water. The molar mass of the anhydrous and the pentahydrate forms of copper sulfate are 159.609 grams/mole and 249.685 grams per mole respectively. Anhydrous Copper sulfate is 39.81 percent copper and 60.19 percent sulfate by mass, and in its blue, hydrous form, it is 25.47% copper, 38.47% sulfate (12.82% sulfur), and 36.06% water by mass. Four types of crystal size are provided based on its usage: large crystals (10–40 mm), small crystals (2–10 mm), snow crystals (less than 2 mm), and windswept powder (less than 0.15 mm).
Uses
- Copper sulfate is used as a fungicide, algaecide, root killer, and herbicide in both agriculture and non-agricultural settings.
- It is also used as an antimicrobial and molluscicide.
- It is used as a drying agent in the anhydrous form, as an additive for fertilizers and foods, and several industrial applications such as textiles, leather, wood, batteries, ink, petroleum, paint, and metal, among others.
- It is used also as an animal nutritional supplement.
Information Source: