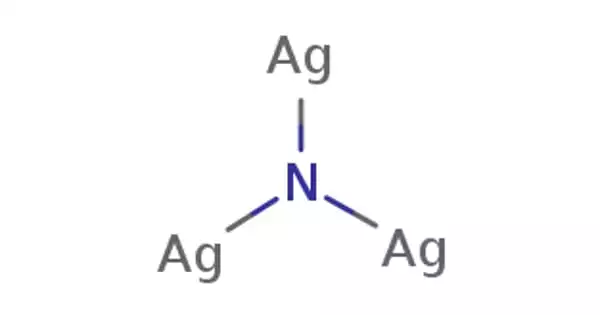
Sodium selenide, also known as Na2Se, is an inorganic compound composed of sodium and selenium. It is a crystalline solid that ranges in color from yellow to red. It is highly reactive and, if handled incorrectly, can be dangerous. It is used as a selenium source in a variety of chemical reactions and synthesis processes. It can be used in the synthesis of other selenium compounds or as a catalyst in certain reactions. It is a good source of selenium, which is a trace element that is required for many biological processes.
Properties
- Chemical formula: Na2Se
- Molar mass: 124.951 g·mol−1
- Density: 2.62 g cm−3
- Melting point: >875 °C
- Solubility in water: reacts with water
- Crystal structure: Cubic (fluorite), cF12
Preparation
This colourless solid is prepared by the reaction of selenium with a solution of sodium in liquid ammonia at −40 °C. Alternatively, sodium selenide can be prepared by the reaction of gaseous hydrogen selenide with metallic sodium at 100 °C. It is soluble in water and forms an alkaline solution.
Reactions
It is a strong reducing agent and can react violently with acids and oxidizing agents. Like other alkali metal chalcogenides, this material is highly sensitive to water, easily undergoing hydrolysis to give mixtures of sodium biselenide (NaSeH) and hydroxide. This hydrolysis occurs because of the extreme basicity of the Se2− ion.
Na2Se + H2O → NaHSe + NaOH
Similarly, sodium selenide is readily oxidized to polyselenides, a conversion signaled by off-white samples.
Sodium selenide reacts with acids to produce toxic hydrogen selenide gas.
Na2Se + 2 HCl → H2Se + 2 NaCl
The compound reacts with electrophiles to produce the selenium compounds. With alkyl halides, one obtains a variety of organoselenium compounds:
Na2Se + 2 RBr → R2Se + 2 NaBr
Uses
Sodium selenide is used to make semiconducting materials such as thin films for electronic devices such as photovoltaic cells, sensors, and rectifiers. It is used in the glass industry to create red glasses. It is sometimes used in research and laboratory settings for specific experiments and reactions due to its reactivity.
Sodium selenide is extremely toxic and should be avoided.