Calcium Chloride
Definition
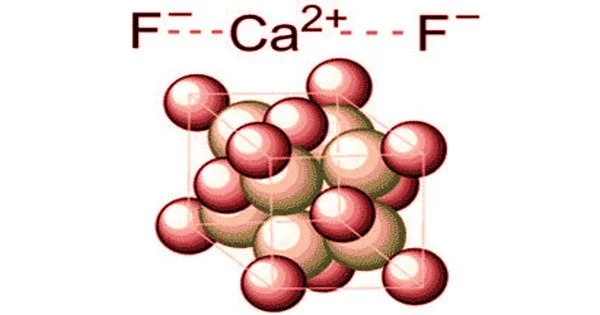
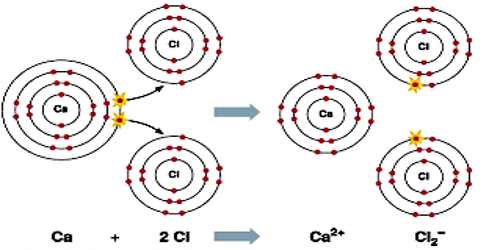
Calcium chloride is a white crystalline salt that attracts water very strongly. It is used in refrigeration and is spread on roads to melt ice and control dust. It is an ionic compound of calcium and chlorine. It is highly soluble in water and it is deliquescent. It is a salt that is solid at room temperature, and it behaves as a typical ionic halide. Its chemical formula CaCl2.

Calcium chloride is commonly encountered as a hydrated solid with generic formula CaCl2(H2O)x, where x = 0, 1, 2, 4, and 6. These compounds are mainly used for de-icing and dust control. Because the anhydrous salt is hygroscopic, it is used as a desiccant.
It has several common applications such as brine for refrigeration plants, ice and dust control on roads, and in cement. It can be produced directly from limestone, but large amounts are also produced as a by-product of the Solvay process. Because of its hygroscopic nature, it must be kept in tightly-sealed containers.
Properties and Functions of Calcium Chloride
Calcium chloride is highly water soluble, hygroscopic (absorbs moisture from air) and deliquescent (absorbs enough water to turn into liquid). Calcium chloride dissolves in water in a very exothermic manner (releasing a large amount of heat). Calcium chloride fully dissociates in water to give calcium cations and chloride anions. In aqueous solutions, the calcium ions can displace other ions in exchange reactions, such as the conversion of potassium phosphate into calcium phosphate:
3 CaCl2 + 2 K3PO4 → Ca3(PO4)2 + 6 KCl
Calcium chloride is found as an odorless white powder, granules or flakes. It has a density of 2.15 g/mL, melting point of 782 °C and a high boiling point over 1600 °C.
Calcium chloride is mainly produced by reacting limestone (CaCO3) with hydrochloric acid (HCl). It occurs as the rare evaporite minerals sinjarite (dihydrate) and antarcticite (hexahydrate). The related minerals chlorocalcite (potassium calcium chloride, KCaCl3) and tachyhydrite (calcium magnesium chloride, CaMg2Cl6•12H2O) are also very rare.
Calcium chloride serves as a firming agent in prepared foods, including canned fruits and vegetables and tofu. These firming properties also help in the production of cheese, according to calcium chloride manufacturer Muby Chemicals. It makes the curds larger and firmer in store-bought milk, and a single teaspoon of calcium chloride solution can treat 2 gallons of milk. Other uses include as an additive to pickle brine, which gives it the salty taste without loading it with sodium, and as an ingredient in sports drinks.
Uses, Effects and Benefits of Calcium Chloride
Calcium chloride has several similar uses as sodium chloride, and it is used as a food additive, food preservative, for de-icing roads in winter, and as brine in refrigeration plants. It is also used as a swimming pool chemical, in water treatment plants, and for desiccating purposes. It also has applications in metallurgy, oil-well drilling, and rubber, paper, dye and paint industries.

The U.S. Food and Drug Administration (FDA) reports that a review of it and other calcium compounds found in food “provides no evidence that suggests possible untoward effects” at the levels at which manufacturers use it. It is now known that calcium chloride actually has several harmful effects on humans and pets alike. Some of those harmful effects include:
- Burns – When calcium chloride comes in contact with the skin it can cause burns, especially when it comes in contact with large quantities of water.
- Respiratory tract irritation – The dust particles from calcium chloride can be inhaled and cause irritation to the respiratory tract. Repeated exposure can be hazardous to your health.
- Irritation of the mouth, throat and gastrointestinal tracts – If ingested by children it can cause severe irritation. It can also cause severe bouts of vomiting and diarrhea. Children should be seen by a physician if they ingest calcium chloride.
- Skin irritation – When calcium chloride comes in contact with the skin it can cause rashes and other skin irritations.
- Disorientation in birds – Birds that ingest calcium chloride can become disoriented, which can result in their inability to fly.
- Defoliating effect on trees – Calcium chloride can cause the leaves on trees to die and fall off.
- Poisonous to dogs – Calcium chloride can be poisonous to dogs. In fact, large enough quantities can lead to kidney damage and even death in dogs. If your dogs should ingest calcium chloride, they should be taken to the vet for medical care.
While most of calcium chloride’s food uses are for preservative and texture purposes, it does provide health benefits in one use: sports drinks. In these, calcium chloride is one of several ingredients that act as electrolytes, ions that conduct electrical activity and help our body maintain fluid balance as well as proper muscle and nerve functions. As an electrolyte, calcium helps maintain bone health and prevent muscle spasms, and chloride helps keep our heartbeat regular. The ACE (American Council on Exercise) recommends drinking a sports drink with electrolytes before exercise sessions lasting longer than an hour.
Reference: