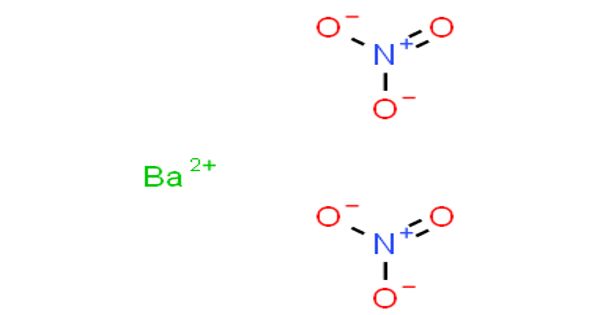
Barium nitrate is the inorganic compound with the chemical formula Ba(NO3)2. It is also called Barium dinitrate or Nitrobarite or Barium salt. It is a salt of barium and a nitrate ion. It, like most barium salts, is colorless, toxic, and water-soluble. It is a good oxidizing agent, burns with a green-colored flame. It burns with a green flame and is an oxidizer; the compound is commonly used in pyrotechnics. It has uses in the vacuum tube industry and the manufacture of other barium compounds. It is widely used in pyrotechnics.
Properties
Barium nitrate appears as a white crystalline solid. If large quantities are involved in fire or the combustible material is finely divided, an explosion may result.
- Molecular weight: 261.337 g/mol
- Density: 3.24 g/dm3
- Refractive index: 1.5659
- Melting Point: 592 °C
- Appearance: White to clear liquid.

Manufacture, occurrence, and reactions
Barium nitrate is manufactured by two processes that start with the main source material for barium, the carbonate. The first involves dissolving barium carbonate in nitric acid, allowing any iron impurities to precipitate, then filtered, evaporated, and crystallized. The second requires combining barium sulfide with nitric acid.
Barium dinitrate is produced by two processes that start with the source component for barium viz carbonate.
Method 1:
- Dissolve barium carbonate (BaCO3) in nitric acid (HNO3)
- Allow the precipitation of iron impurities
- Separate the impurities by filtration
- Evaporation
- Crystallization.
Method 2:
It is obtained by combining barium sulfide (BaS) with nitric acid (HNO3). At higher temperatures, barium nitrate (Ba(NO3)2) decomposes to barium oxide (BaO).
It occurs naturally as the very rare mineral nitrobarite. At elevated temperatures, barium nitrate decomposes to barium oxide:
2Ba(NO3)2 → 2BaO + 4NO2 + O2
Uses
Barium nitrate is used in the production of barium oxide containing materials. Solutions of sulfate salts such as Epsom salts or sodium sulfate may be given as first aid for barium poisoning, as they precipitate the barium as the insoluble (and non-toxic) barium sulfate.
- It is used in green signal lights.
- It is used in making ceramic glazes.
- It is used in detonators.
- It is used in the making of paints.
- It is used as an oxidizing agent.
- It is used in making explosives.
Safety
It is a non-combustible compound, but enhances the burning of combustible elements. Like all soluble barium compounds, barium nitrate is toxic by ingestion or inhalation. When exposed to fire or heat for a long duration it may explode.
Inhalation may also cause irritation to the respiratory tract. While skin or eye contact is less harmful than ingestion or inhalation, it can still result in irritation, itching, redness, and pain.
The Occupational Safety and Health Administration and the National Institute for Occupational Safety and Health have set occupational exposure limits at 0.5 mg/m3 over an eight-hour time-weighted average.
Information Source: