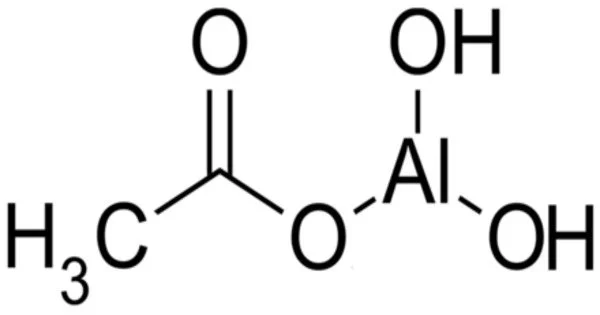
Aluminium monoacetate, also known as dibasic aluminium acetate, and formally named dihydroxy aluminium acetate, is a salt of aluminium with acetic acid. It has the formula Al(OH)2(CH3COO), with aluminium in an oxidation state of +3, and appears under standard conditions as a white solid powder. It forms from Al(OH)3 and dilute aqueous acetic acid. More concentrated acid leads to the di- and triacetate.
It is widely used as an antiseptic agent. It is used to treat inflammation, stinging of the infected skin, itching, and promotes healing.
Properties
- Chemical formula: (HO)2AlCH3CO2 or C2H5AlO4
- Molar mass: 120.04 g/mol
- Appearance: White powder
Chemistry
Aluminium monoacetate is prepared from the reaction between Al(OH)3 and dilute aqueous acetic acid. It is also formed from the successive hydrolysis of aluminium triacetate.
Al(CH3COO)3 + H2O → Al(OH)(CH3COO)2 + CH2COOH
Al(OH)(CH3COO)2 + H2O → Al(OH)2(CH3COO) + CH3COOH
Uses
Aluminium monoacetate is a dermatological agent that acts as an antiseptic and astringent. It is used as an antiseptic to reduce the possibility of infection in minor wounds, cuts, and burns.
It specifically treats itching, stinging of infected skin, inflammation, and promotes healing. It can also be used as a topical astringent to help shrink the body’s tissues when applied to the skin, as it acts as a protective layer on irritated and inflamed skin.