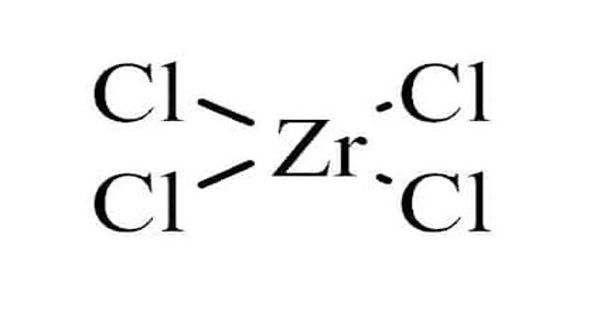Zirconium(IV) chloride forms lustrous, white, monoclinic crystals that melt at 437℃. It is a corrosive powder. It is also known as zirconium tetrachloride, (ZrCl4) is an inorganic compound frequently used as a precursor to other compounds of zirconium. It is commonly utilized as a Lewis acid catalyst in many organic synthesis reactions. This white high-melting solid hydrolyze rapidly in the humid air. It is hygroscopic, and thus soluble in cold water, alcohol, ether, and concentrated hydrochloric acid.
It is an important source for organometallic complexes of zirconium. It is practiced in many areas like the nuclear industry, metal industry, photography, glass, and ceramic industry.
Properties
Hydrolysis of ZrCl4 gives the hydrated hydroxy chloride cluster called zirconyl chloride. This reaction is rapid and virtually irreversible, consistent with the high oxophilicity of zirconium(IV). For this reason, manipulations of ZrCl4 typically require air-free techniques.
It is stable under room temperatures. Strongly hygroscopic and decomposes in water forming ZrOCl2 and HCl.
- Compound Formula: Cl4Zr
- Molecular Weight: 233.04
- Appearance: white crystals
- Melting Point: 437°C (818.6°F)
- Boiling Point: 331°C (627.8°F)
- Density: 2.8 g/cm3
- Solubility in H2O: N/A
ZrCl4 is the principal starting compound for the synthesis of many organometallic complexes of zirconium. Because of its polymeric structure, ZrCl4 is usually converted to a molecular complex before use.
Structure
Unlike molecular TiCl4, solid ZrCl4 adopts a polymeric structure wherein each Zr is octahedrally coordinated. This difference in structures is responsible for the disparity in their properties: TiCl4 is distillable, but ZrCl4 is solid. In the solid state, ZrCl4 adopts a tape-like linear polymeric structure—the same structure adopted by HfCl4. This polymer degrades readily upon treatment with Lewis bases, which cleave the Zr-Cl-Zr linkages.
Synthesis
This conversion entails the treatment of the oxide with carbon as the oxide “getter” and chlorine.
ZrO2 + 2 C + 2 Cl2 → ZrCl4 + 2 CO
A laboratory-scale process uses carbon tetrachloride in place of carbon and chlorine:
ZrO2 + 2 CCl4 → ZrCl4 + 2 COCl2
It reacts vigorously with water forming hydrogen chloride (irritating vapor) and zirconium oxychloride. When moist, zirconium tetrachloride reacts with common materials to form hydrochloric acid that is corrosive to many metals.
Uses:
- Activates pyrrolidines for improved conversion, via a modified Bouveault reaction, to the corresponding α,α-dimethylamines.
- Zirconium (IV) chloride is used as an efficient catalyst in the Pechmann condensation reaction of phenols with β‐keto esters leading to the formation of coumarin derivatives in good yields under solvent‐free conditions.
Information Source: