Human immunodeficiency virus (HIV) is the virus that causes acquired immunodeficiency syndrome (AIDS), a disease characterized by progressive immune system deterioration that eventually leads to death, usually from cancer or infection. According to Duke Health researchers, an immune response that likely evolved to help fight infections appears to be the mechanism that drives human immunodeficiency virus (HIV) into a latent state, lurking in cells only to erupt again.
The research team published their findings in the journal Nature Microbiology, providing new insights into the perplexing process that not only makes HIV particularly stealthy but may also play a role in other viral infections.
“HIV has proven to be incurable because of a small number of latently HIV-infected T-cells that are unaffected by both antiviral drugs and the immune response,” said senior author Bryan R. Cullen, Ph.D., professor in Duke University School of Medicine’s Department of Molecular Genetics and Microbiology.
“These cells, which have a very long life span, can spontaneously emerge from dormancy and begin producing HIV even years after infection, necessitating lifelong antiretroviral therapy,” Cullen explained. “Despite considerable effort, the origin of these latently infected cells has remained unknown.”
Our findings suggest that latency is caused by a cellular innate immune response that evolved to silence invasive foreign DNA rather than any intrinsic properties of the infecting HIV.
Bryan R. Cullen
The findings from Cullen and colleagues offer important insights, pointing to a protein complex called SMC5/6, which is involved in a host cell’s chromosome function and repair.
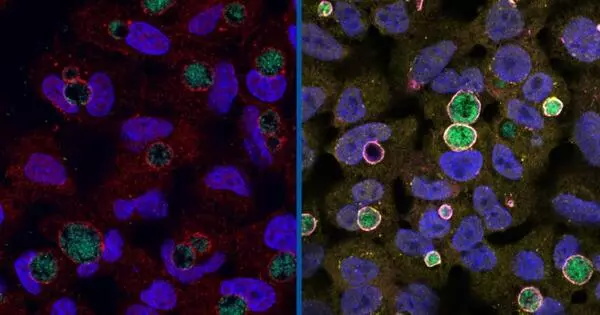
HIV enters the body, infects the immune system’s CD4+ T-cells, then makes a genome-length DNA molecule that it integrates into a host cell chromosome where it is then copied to generate viral RNAs and proteins.
If this so-called DNA provirus is prevented from integrating into the host cell DNA, for example by a drug that blocks this process, then it fails to make any viral RNAs and proteins and becomes inert. In contrast, DNA proviruses that are able to integrate are normally able to drive a productive HIV infection.
Cullen and his colleagues discovered that the SMC5/6 protein complex initiates a process that silences the DNA provirus before it integrates into a host cell chromosome in a small number of infected cells. Even after integration, these proviruses remain dormant, resulting in latent infections that remain dormant until triggered into active infections.
“Our findings suggest that latency is caused by a cellular innate immune response that evolved to silence invasive foreign DNA rather than any intrinsic properties of the infecting HIV,” Cullen said.
The researchers found that a molecule that shuts down SMC5/6’s silencing action showed promising results as a potential therapeutic strategy as it inhibited the establishment of latent HIV infections. Reactivated proviruses are vulnerable to natural immune system responses and anti-retroviral drugs.
“While antiretroviral therapies can reduce viral load in AIDS patients to levels below detection, these drugs do not eradicate HIV-1,” Cullen explained. “While considerable effort has been expended in attempting to develop therapies that can activate latent HIV-1 and aid antiretroviral therapies in clearing the body of the infectious virus, this effort has so far failed to identify drugs that are both effective and non-toxic. Our research could be a significant step toward achieving this goal.”
“Understanding the mechanism that causes HIV-1 latency may provide insights into how latent HIV-1 proviruses can be reactivated and then destroyed,” Cullen said.