The ability of certain chemical compounds to store energy in the form of chemical bonds is referred to as energy storage in molecules. When necessary, these molecules can release the stored energy through a variety of chemical events. This idea is critical in biology, chemistry, and technology since it is critical in the energy metabolism of living organisms and the creation of energy storage technologies.
Molecular photoswitches that can convert and store energy could be utilized to improve the efficiency of solar energy harvesting. A group of scientists employed quantum computing to identify a particularly efficient chemical structure for this function. As the team described in the journal Angewandte Chemie, their procedure was based on a dataset of more than 400,000 molecules, which they screened to find the optimum molecular structure for solar energy storage materials.
Solar energy is currently used either directly to generate electricity or indirectly through the energy stored in heat reservoirs. A third option would be to store solar energy in light-sensitive materials and then release it as needed. The EU-funded project MOST (“Molecular Solar Thermal Energy Storage”) is investigating molecules such as photoswitches that can absorb and store solar energy at room temperature in order to make completely emission-free solar energy use a reality.
While the systems can be synthesized, there is no guarantee that they are soluble in relevant solvents and that they will actually photoswitch in high yield or at all, as we assumed in eta.
Kurt V. Mikkelsen
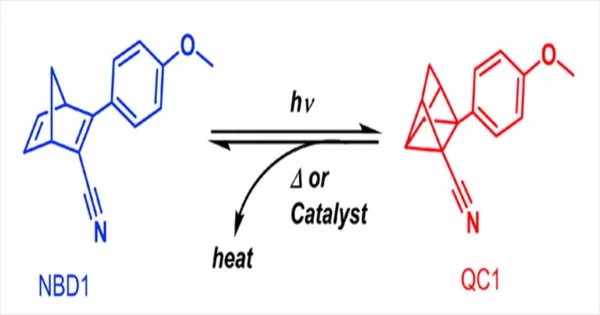
The research teams of Kurt V. Mikkelsen at the University of Copenhagen, (Denmark) and Kasper Moth-Poulsen at the Technical University of Catalonia, Barcelona (Spain), have taken a closer look at the photoswitches best suited for this task. They studied molecules known as bicyclic dienes, which switch to a high-energy state when illuminated. The most prominent example of this bicyclic diene system is known as norbornadiene quadricyclane, but a vast number of similar candidates exist.
According to the researchers, “the resulting chemical space consists of approximately 466,000 bicyclic dienes that we have screened for their potential applicability in MOST technology.”
Machine learning is generally used to screen a database of this scale, but this requires vast quantities of training data based on real-world experiments, which the team lacked. The screening and evaluation of the database molecules using a previously created algorithm and a novel evaluation score, “eta,” gave a clear result: all six of the top-scoring molecules deviated from the original norbornadiene quadricyclane system at a critical place in the structure.
The researchers determined that the structural modification, which involved an increase of the chemical bridge between the two carbon rings in the bicyclic section, enabled the new molecules to store more energy than the original norbornadiene.
The work of the researchers reveals the possibility of optimizing solar energy storage molecules. The new compounds, however, must first be manufactured and evaluated under real-world situations. “While the systems can be synthesized, there is no guarantee that they are soluble in relevant solvents and that they will actually photoswitch in high yield or at all, as we assumed in eta,” the scientists warn.
Despite this, the researchers created a fresh, vast amount of training data for machine learning algorithms, shortening the hard research stage prior to synthesis for future chemists confronting such systems. The scientists see this much larger collection of bicyclic dienes coming into its own for research into photoswitches for a range of applications, potentially making it easier to customize molecules to specific needs.