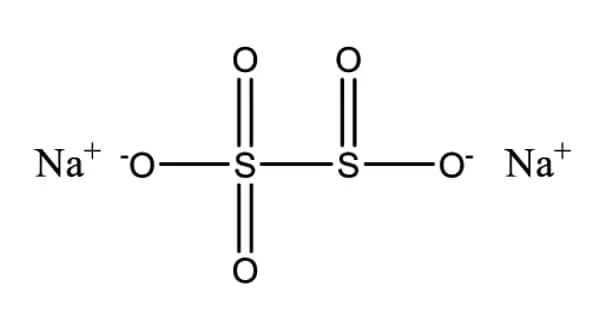
Sodium metabisulfite, also known as sodium pyrosulfite, is an inorganic compound with the formula Na2S2O5. It has a slight sulfur odor and appears as a white crystalline or powder solid. The substance is sometimes referred to as disodium metabisulfite. It is a reducing agent that is commonly used for the preservation of fresh & dried fruits, vegetables, and wines. It is also used as a disinfectant, antioxidant, and preservative agent.
Properties
It is white in color, solid, and powder form. It has a similar odor to Sulfur dioxide. The molar mass of sodium metabisulfite is 190.107 g/mol. It appears as a white crystalline or powder solid with a slight sulfur odor.
- Molar Mass: 190.107 grams per mole
- Density: 1.48 grams per cubic centimetre
- Melting Point: 170oC but begins to decompose at 150oC
- Boiling Point: Decomposes
- Storage temp.: Store at +5°C to +30°C.
- Solubility 540 g/L (20°C)
- Form: Powder/Solid
- Color: White to slightly yellow
- Specific Gravity: 1.48

Preparation
Sodium disulfite can be prepared by treating a solution of sodium hydroxide with sulfur dioxide. When conducted in warm water, Na2SO3 initially precipitates as a yellow solid. With more SO2, the solid dissolves to give the disulfite, which crystallizes upon cooling.
SO2 + 2 NaOH → Na2SO3 + H2O
SO2 + Na2SO3 → Na2S2O5
which yields a residue of colorless solid Na2S2O5.
Here is the manufacturing process that came up by Solvay: Reaction between sodium carbonate (Na2CO3) and sodium bisulfite (NaHSO3) solution to form the mixed solution of sodium bisulfite and sodium sulfite (Na2SO3). Reaction equation:
Na2CO3 + 2 NaHSO3 = Na2SO3 + H2O+CO2
Add excess sulfur dioxide (SO2) to the above solution to convert sodium sulfite to sodium bisulfite and also obtain the dissolved sulfur dioxide. Reaction equation:
Na2SO3 + H2O + SO2 = 2NaHSO3
Convert the dissolved sulfur dioxide to sodium bisulfite by adding an alkali ingredient, e,g, sodium carbonate, sodium hydroxide, or sodium sulfite, or the mixtures.
Uses
Many major and minor applications exist for sodium and potassium metabisulfite. It is commonly used to preserve food and beverages. To prevent adrenaline oxidation, sodium metabisulfite is added as an excipient to medications containing adrenaline (epinephrine). It is added, for example, to combination drug formulations containing a local anesthetic and adrenaline, as well as to the formulation in epinephrine autoinjectors such as the EpiPen. Although the sodium metabisulphite reacts with adrenaline, causing it to degrade and form epinephrine sulphonate, this extends the shelf life of the formulation.
- It is used to remove rust stains when combined with sodium hydrosulfite.
- It is employed in the field of photography.
- Tree stumps can be removed with concentrated sodium metabisulfite. Some brands contain 98 percent sodium metabisulfite, which causes lignin degradation in the stumps, allowing for easier removal.
- It’s also used as an excipient in some medications, like paracetamol.
- This compound is also used in the treatment of wastewater since it can reduce hexavalent chromium into its trivalent form.
Precaution
It can be produced by evaporating a sodium bisulfite solution that has been saturated with sulfur dioxide. It should be noted that sodium metabisulfite has some serious side effects on the respiratory system, eyes, and skin. It can cause breathing difficulties and even pulmonary damage in severe cases, eventually leading to death. As a result, effective precautions and attention should be taken during the operation.