Reference Electrode is an electrode having an accurately maintained potential, used as a reference for measurement by other electrodes. The high stability of the electrode potential is usually reached by employing a redox system with constant (buffered or saturated) concentrations of each participant of the redox reaction. Electrodes are vital components of electrochemical cells. They transport produced electrons from one half-cell to another, which produces an electrical charge.
A reference electrode is an electrode which has a stable and well-known electrode potential. There are many ways reference electrodes are used. Its characteristics can greatly influence electrochemical measurements. The simplest is when the reference electrode is used as a half-cell to build an electrochemical cell. The electrode can either be an anode or a cathode. An anode receives current or electrons from the electrolyte mixture, thus becoming oxidized. This allows the potential of the other half cell to be determined. An accurate and practical method to measure an electrode’s potential in isolation (absolute electrode potential) has yet to be developed.
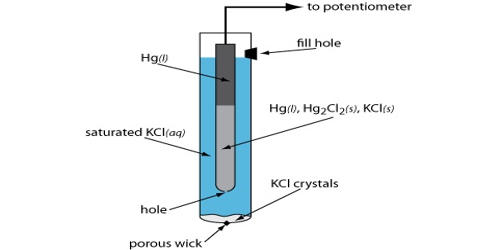
Reference electrodes are used as the third electrode in three-electrode electrochemical cell systems for cyclic voltammetry and similar studies. For a reference electrode to be useful, it must have both a stable and reproducible potential to which the indicating electrode potential can be compared. They are sometimes referred to simply as the third electrode. Importantly, reference electrodes have a stable, known, and well-defined electrochemical potential. Reference electrodes are commonly used with a saturated solution and an excess of the salt crystals. The extra salt dissolves into the half-cell solution as some of the ions diffuse out of the reference cell body through the liquid junction during normal use.
Everyone agrees that an ideal reference electrode has a stable, well-defined electrochemical potential. Reference electrodes should have stable and reproducible potential. Common reference electrodes (SCE, Ag/AgCl, Cu/CuSO4) meet this criterion when they are functioning properly. These electrodes used are preferably reversible type electrodes. Many workers do not realize how often reference electrodes fail, causing drastic changes in their potential. In a reversible electrode, a small cathodic current produces the reduction reaction, while a small anodic current produces the oxidation reaction. Many complaints of potentiostat failures turn out to be reference electrode failures. An ideal reference electrode should also have zero impedance. This type of electrode is durable, reliable, and commercially available. The potential of a reference electrode should be checked at periodic intervals to ensure the accuracy of the reference electrode.