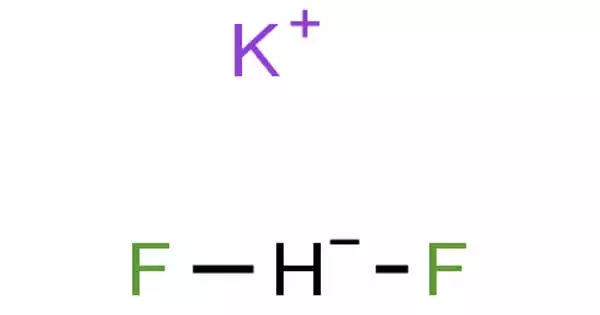The inorganic compound with the formula KHF2 is potassium bifluoride. This colorless salt is made up of potassium cation and bifluoride (HF2) anion. The salt is used in glass etchant. Sodium bifluoride is related to it and is also used commercially as an etchant and in cleaning products.
Potassium bifluoride is widely used in the production of soldering agents and wood preservatives, as a catalyst in condensation and polymerization in the chemical industry, in the production and treatment of glass, and for surface treatments.
Properties
Potassium bifluoride appears as colorless crystals, corrosive to tissues, etches glass. This colorless salt consists of the potassium cation and the bifluoride anion. The salt is used in the etchant for glass.
- Molecular Weight: 78.11
- Appearance: Colorless Hygroscopic Crystals
- Melting Point: 238.9 °C
- Boiling Point: N/A
- Density: 2.37 g/cm3
- Solubility in H2O: Soluble in H2O; Insoluble in EtOH
- Heat of Fusion: 84.76 J/g

Synthesis and reactions
The salt was prepared by Edmond Frémy by treating potassium carbonate or potassium hydroxide with hydrofluoric acid:
2 HF + KOH → KHF2 + H2O
With two more equivalents of HF, KH2F3 is produced:
2 HF + KHF2 → KH2F3
Thermal decomposition of KHF2 gives hydrogen fluoride:
KHF2 → HF + KF
Production
Potassium bifluoride is manufactured by mixing anhydrous hydrogen fluoride (AHF, liquid) and potassium hydroxide, and then drying to form a powder.
2HF + KOH → KHF2 + H2O
Applications
The industrial production of fluorine entails the electrolysis of molten KHF2 and KH2F3. The electrolysis of KHF2 was first used by Henri Moissan in 1886. It is used in the chemical industry, manufacture of wood preservatives glass processing, glass manufacturing, and for the production of soldering agents. It is used in the following applications:
- Glass manufacturing: for special optical glasses (Crown and Crown Flint glass)
- Glass treatment: for matt etching
- Wood preserving agents: as active component for wood preservatives
- Production of soldering agents: as component of fluxing agents for soldering
- Chemical industry: for manufacturing of organic and inorganic fluorine compounds.
Safety
Potassium bifluoride is a corrosive chemical that can irritate and burn the skin and eyes, causing permanent eye damage. Breathing in potassium bifluoride dust can irritate and burn the nose, throat, and lungs, resulting in nosebleeds, coughing, wheezing, and shortness of breath. When KBF comes into contact with water or moist skin, it can produce hydrofluoric acid, a highly toxic acid. When potassium bifluoride is heated, it emits hydrogen fluoride, a toxic and corrosive gas.
Large amounts of potassium bifluoride inhaled or ingested can cause nausea, vomiting, and loss of appetite. Fluoride poisoning can result from high concentrations or long-term exposure, and symptoms include stomach pain, weakness, convulsions, and death. Fluoride deposits in bones and teeth can result from long-term or repeated exposures, a condition known as fluorosis. Fluorosis may cause pain, disability and discoloration of teeth.