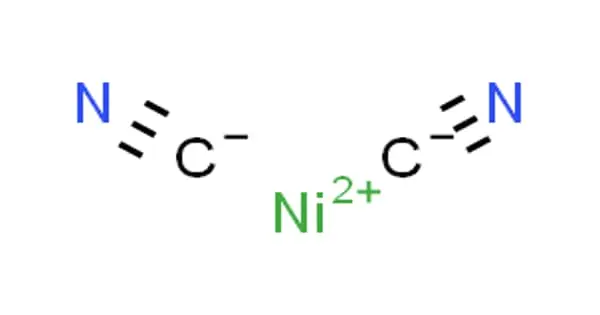
Nickel dicyanide is an inorganic compound with a chemical formula Ni(CN)2. It is a gray-green solid that is insoluble in most solvents. It is an apple-green powder or a green crystalline solid. It is insoluble in water. It is toxic by inhalation and by ingestion. It produces toxic oxides of nitrogen in fires.
Production
The addition of two equivalents of sodium or potassium cyanide to a solution of nickel(II) ions in an aqueous solution leads to the precipitation of nickel(II) cyanide tetrahydrate. On heating the tetrahydrate to 140 °C, this hydrate converts to anhydrous nickel(II) cyanide.
Structure
The structure of the nickel cyanide ammoniate complex has been determined by x-ray diffraction methods. The cell is tetragonal with a = 7.222 Å and o = 17.42 Å. The nickel and cyanide atoms pack in extended square arrays perpendicular to the c axis.
Chemical properties
Nickel(II) cyanide dissolves in potassium cyanide solution to produce a yellowish solution containing potassium tetracyanonickelate:
Ni(CN)2 + 2 KCN → K2[Ni(CN)4]
Nickel(II) cyanide will react with dimethylglyoxime (dmgH2) and produce hydrogen cyanide:
Ni(CN)2 + 2 dmgH2 → Ni(dmgH)2 + 2 HCN
Preparation
Nickel cyanide is prepared by treating a soluble nickel salt, such as nickel chloride or nickel sulfate, with potassium cyanide solution: Ni2+ + 2CNˉ → Ni(CN)2
The product is a tetrahydrate, Ni(CN)2.4H2O, which on heating at 200°C yields yellow-brown anhydrous salt, Ni(CN).
Reactions
The most interesting reaction of nickel(II) cyanide is the formation of clathrate compounds in the presence of ammonia. When a solution of Ni(CN)2 in aqueous ammonia is shaken with benzene, a pale violet precipitate of the benzene clathrate is obtained. In this compound, the Ni and CN groups form layers with ammonia molecules bonded above and below the planes of the layers on alternate nickel atoms.
Thus half the nickel atoms are octahedrally surrounded by nitrogen (Ni-N=2.15, N1-NH3 = 2.06 à) and half are planar 4-coordinated by carbon (Ni-C=1.76). The benzene molecules occupy the holes (cages) formed between the layers. The average magnetic moment per nickel atom is 2.2 BM, and it has thus been established that the 4- coordinate nickel atoms are diamagnetic while the 6-coordinate atoms are paramagnetic with two unpaired electron spins.