Plastic bags and gasoline are two products that environmentalists frequently urge the public to abandon for the sake of the planet. Scientists believe they have discovered a way to convert plastic bags into a more sustainable source of fuel. California State Polytechnic University researchers developed a method for chemically converting plastic waste into a diesel-like substance.
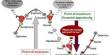
The researchers used catalytic pyrolysis to convert plastic waste into a valuable fuel source. They concentrated on recycling plastic and upgrading it into other products, as well as converting it to vapor with heat, which met a catalyst and transformed into the desired fuel-like product. This pyrolytic process converts primary organic waste into a long-lasting fuel or other valuable chemicals.
More than 300 million tons of plastic waste are produced each year, causing serious environmental issues due to the life cycle of plastic and the difficulty in eliminating it. According to estimates, humans generate approximately 300 million tons of plastic waste each year. To make matters worse, the life cycle of plastic makes it difficult to remove these products from the environment once they enter landfills.
Many of these plastics eventually degrade into microplastics, which enter the ocean and other ecosystems. Fish and other animals then consume these particles, which have an impact on their own health and can even disrupt human health when people consume seafood and other products.
As a result, the majority of plastic waste ends up in a landfill or the ocean. Many plastics degrade into microplastics, which are consumed by fish and other marine life, wreaking havoc on marine ecosystems.
The catalyst is the innovative part of the experiment. The catalyst is critical to this specific pyrolysis process because it only requires one step to get to the desired fuel product at relatively low temperatures.
Mingheng Li
Researchers from California State Polytechnic University report in AIP Publishing’s Journal of Renewable and Sustainable Energy that they used catalytic pyrolysis to convert plastic waste into a valuable fuel source. The thermochemical decomposition of carbon-based matter in the absence of oxygen is known as pyrolysis.
Researchers concentrated on recycling plastic and upgrading it into other products, as well as converting it to vapor with heat, which met a catalyst and transformed into the desired fuel-like product. This pyrolytic process converts primary organic waste into a long-lasting fuel or other valuable chemicals.
Scientists developed their catalyst by immersing a zeolite substrate (a mineral) in a nickel and tungsten solution. The substance was then dried in an oven at over 900 degrees Fahrenheit. They used this catalyst in conjunction with a single-stage pyrolytic reactor operating at nearly 700 degrees Fahrenheit to degrade common plastic grocery bags.

“The catalyst is the innovative part of the experiment,” said author Mingheng Li. “The catalyst is critical to this specific pyrolysis process because it only requires one step to get to the desired fuel product at relatively low temperatures.”
The catalyst was created by immersing a zeolite substrate in an aqueous solution containing nickel and tungsten and drying it in a 500°C oven. The synthesized catalyst was combined with a lab-designed, single-stage pyrolytic reactor that ran at a set temperature of 360 C to degrade a mixture of plastic grocery bags.
The catalytic process used in this experiment on plastic waste could be used to convert other wastes, such as manure, municipal solid waste, and used engine oil, into usable energy products. “This pyrolysis process is a decisive step toward reducing reliance on fossil-based fuels,” Li said.
Using gas chromatographic analysis, the researchers discovered that the pyrolysis product was very similar to a standard diesel fuel product. Gas chromatography is a type of chromatography used in analytical chemistry for separating and analyzing compounds that can be vaporized without decomposition. The team will now work to explain the cracking mechanism that occurs on the catalyst’s surface. They will also try to optimize the production of diesel fuel from various mixed plastic wastes.
The facilities involved in the plastic-to-fuel transformation have the potential to create up to 39,000 new jobs and nearly $9 billion in economic output. This has the potential to boost our economy while also providing new ways to reuse plastic and save the environment.
This new method may also be less expensive than current recycling methods. Currently, it costs upwards of $4,000 to recycle one tonne of plastic bags; to avoid these high costs, many people burn plastic or dispose of it in landfills.