Lithium aluminate (LiAlO2), also known as lithium aluminium oxide, is an inorganic chemical compound that is a lithium aluminate. Lithium aluminate is used as a lattice matching substrate for gallium nitride in microelectronics. Lithium aluminate is of interest in nuclear technology as a solid tritium breeder material for preparing tritium fuel for nuclear fusion.
Properties
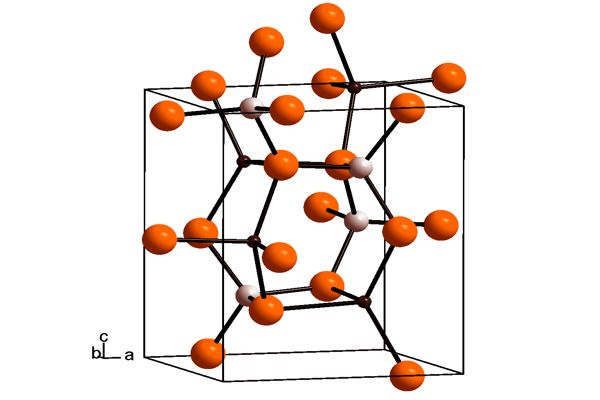
Lithium aluminate is a layered double hydroxide (LDH) with a crystal structure resembling that of hydrotalcite. Lithium aluminate solubility at high pH (12.5 – 13.5) is much lower than that of aluminium oxides.
- Molecular Weight: 65.921
- Appearance: White crystalline powder or solid in various forms such as rod
- Melting Point: 1625 °C
- Boiling Point: N/A
- Density: 2.615 g/cm3
- Solubility in H2O: Insoluble
- Exact Mass: 65.987372
- Monoisotopic Mass: 65.987372
Lithium nitrate is sometimes used as an additive to cement in the conditioning of low- and intermediate-level radioactive waste (LILW) to reduce aluminum corrosion at high pH and subsequent hydrogen production. When lithium nitrate is added to cement, a passive layer of LiH(AlO2)2 • 5 H2O forms on the surface of metallic aluminum waste immobilized in mortar.
Solubility
The lithium aluminate layer is insoluble in cement pore water and protects the underlying aluminum oxide layer, which covers the metallic aluminum, from dissolution at high pH. It also acts as a pore filler. This inhibits the oxidation of aluminum by water protons and reduces the rate of hydrogen evolution by a factor of ten.
Application
- It is used as an inert electrolyte support material in molten carbonate fuel cells.
- A lithium aluminate – MgO catalyst has been found to convert methane to ethylene with high selectivity.
- Lithium aluminate also finds its use as an inert electrolyte support material in molten carbonate fuel cells, where the electrolyte may be a mixture of lithium carbonate, potassium carbonate, and sodium carbonate.
Information Source: