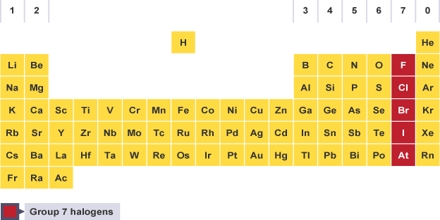
The halogens are located on the left of the noble gases on the periodic table. These five toxic, non-metallic elements make up Group 17 of the periodic table and consist of: fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At). Although astatine is radioactive and only has short-lived isotopes, it behaves similar to iodine and is often included in the halogen group. Because the halogen elements have seven valence electrons, they only require one additional electron to form a full octet.
The Halogens