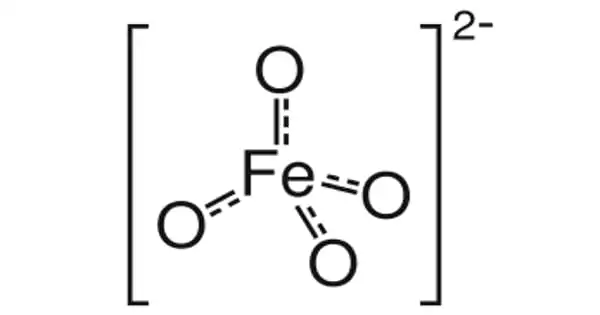
The chemical compound barium ferrate has the formula BaFeO4. It is a barium and iron chemical compound. This is a rare iron compound with a +6 oxidation state. The ferrate (VI) ion is paramagnetic because it has two unpaired electrons. It is structurally equivalent to BaSO4 and contains the tetrahedral [FeO4]2- anion.
Barium is a metallic alkaline earth metal with the atomic number 56 and the symbol Ba. Due to its reactivity with air, it never occurs in its pure form in nature, but instead combines with other chemicals such as sulfur or carbon and oxygen to form barium compounds, which can be found as minerals.
Properties
It is a pinkish-brown powder that is insoluble in water and other solvents. It is a strong oxidizer, but because of its insolubility, it is extremely stable for ferrate. It is not known to decompose simply by coming into contact with air, as potassium ferrate frequently does. This is a stable barium salt of ferric acid, which is too unstable to exist in water.
- Molecular Weight: 1111.46
- Appearance: Gray to black powder
- Melting Point: 1316 °C
- Boiling Point: N/A
- Density: 5.28 g/cm3
- Solubility in H2O: Insoluble

Preparation
Both wet and dry synthetic methods can be used to produce barium ferrate(VI). Dry synthesis is typically carried out using a thermal technique, such as heating barium hydroxide and iron(II) hydroxide in the presence of oxygen to temperatures ranging from 800 to 900°C.
Ba(OH)2 + Fe(OH)2 + O2 → BaFeO4 + 2 H2O
Chemical and electrochemical techniques. When a suitable iron salt is placed in alkaline conditions and a strong oxidizing agent, such as sodium hypochlorite, is added, the ferrate anion forms.
2 Fe(OH)3 + 3 OCl– + 4 OH– → 2 FeO42- + 5 H2O + 3 Cl–
The precipitation of barium ferrate from the solution is then accomplished by adding a solution of barium (II) salt. When a soluble barium salt is added to an alkali metal ferrate solution, a maroon precipitate of barium ferrate is formed, a crystal with the same structure as barium chromate and approximately the same solubility. At room temperature (or 0 °C), barium ferrate was also prepared by adding barium oxide to a mixture of sodium hypochlorite and ferric nitrate. The purity of the product can be increased by performing the reaction at a low temperature in the absence of carbon dioxide and rapidly filtering and drying the precipitate, which reduces the coprecipitation of barium hydroxide and barium carbonate as impurities.
Uses
Barium ferrate is an oxidizing agent that is used in organic synthesis as an oxidizing reagent. Other applications include color removal, cyanide removal, bacteria-killing, and contaminated and wastewater treatment.
Ferrate(VI) salts are active cathode materials in “super-iron” batteries. Because of their highly oxidized iron basis, multiple electron transfer, and high intrinsic energy, cathodes containing ferrate(VI) compounds are referred to as “super-iron” cathodes. Among all ferrate(VI) salts, barium ferrate has an unusually easy charge transfer, which is important for alkaline batteries’ high power domain.