Cellular waste disposal is a critical process that ensures the maintenance of cellular homeostasis and cell function. The cellular structures known as lysosomes are at the heart of this waste disposal system. Lysosomes are membrane-bound organelles found in animal and plant cells that contain enzymes capable of degrading and recycling cellular waste. Researchers explain how a nanomachine cleans up inside the cell.
To keep our bodies’ cells from overflowing with garbage and to keep them healthy, waste is constantly disposed of within them. This cleaning procedure is known as autophagy. For the first time, scientists have rebuilt the complex nanomachine that initiates this process in the laboratory – and it works quite differently than other cellular machines. The researchers’ new findings could pave the way for new approaches to cancer, immune disorders, and neurodegenerative diseases in the future, and may even help to delay aging.
Have you ever put off cleaning your house or organizing your overflowing basement? When it comes to clearing the decks, living cells cannot afford to delay. There are tiny garbage chutes that are constantly active, capturing worn-out proteins, faulty cell components, or faulty organelles. These garbage chutes, known as autophagosomes, remove discarded components from the cell before they accumulate and cause damage. The cellular waste is then transferred to the lysosome, the cell’s own recycling machinery, where it is digested and recycled. As a result, building blocks for new cellular components are readily available. The autophagy process, which literally means “self-eating,” thus aids cells in surviving stress or starvation.
Autophagy is a highly complex process involving many different proteins and protein complexes. We know many of them, but there are still fundamental gaps in our knowledge.
Alex Faesen
Autophagy also serves another important purpose. It renders harmless viruses and bacteria that successfully bypass the immune system’s defenses and reach the cell plasma. The consequences are correspondingly fatal if the autophagy process is faulty, too slow, or too fast. Neurodegenerative diseases and cancer can develop or disorders of the immune system may occur. Aging processes also appear to accelerate.
“Autophagy is a highly complex process involving many different proteins and protein complexes. We know many of them, but there are still fundamental gaps in our knowledge,” reports Alex Faesen, research group leader at the Max Planck Institute for Multidisciplinary Sciences in Göttingen. “How do the protein components work together? How is the process of autophagy started and stopped? When and where is the autophagosome assembled? That is what we want to find out.”
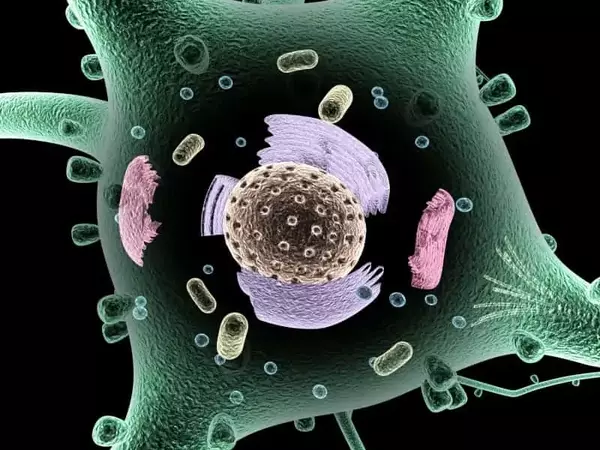
Nanomachine at work
His team has now succeeded in producing all of the proteins involved in the autophagy process in the laboratory and directly observing them as the autophagosomes assemble for the first time. This was a massive task that took several years for the entire research group, and they collaborated with teams led by Björn Stork from the University of Düsseldorf and Michael Meinecke, previously at the University Medical Center Göttingen and now at the Heidelberg University Biochemistry Center.
“There were many challenges,” Faesen recalls. The scientists first created each individual protein component in the laboratory. The standard approach is to use bacteria that have been genetically reprogrammed to produce large amounts of the desired protein. “But protein production with bacteria did not work for any of our proteins,” says the Göttingen biochemist. The breakthrough came when the researchers switched to insect cells as molecular helpers.
The team then brought the individual protein complexes together. “The complexes self-assembled into the autophagy initiation complex, a protein supercomplex.” “In fact, autophagy involves a sophisticated cellular nanomachine that operates quite differently than previously thought,” says the group’s leader.
The autophagy initiation complex first forms a junction between a specific cell structure, the endoplasmic reticulum, and the autophagosome that forms. This happens quickly under stress or in times of starvation, such as during endurance sports.
“From this point on, there is no turning back: the waste disposal is assembled and collects the cellular waste,” says Anh Nguyen, one of the study’s two first authors. Fancesca Lugarini, co-first author, adds, “Via the contact site, fat-like molecules called lipids are transported to a precursor stage of autophagosomes, where they are incorporated.” These develop and enclose the cell material to be discarded, resulting in the finished mini-organelle. Within barely 20 minutes of its formation, the autophagosome is already delivering its waste to the lysosome by fusing with it.