Adiponectin Hormone
Definition
Adiponectin Hormone secreted by adipose tissue that seems to be involved in energy homeostasis; it enhances insulin sensitivity and glucose tolerance, as well as oxidation of fatty acids in muscle. It is involved in regulating glucose levels as well as fatty acid breakdown.
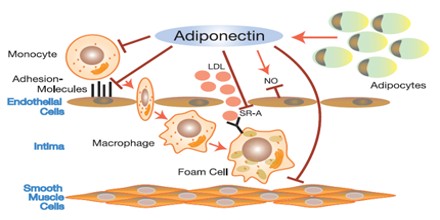
Adiponectin influences the body’s response to insulin. Adiponectin also has antiinflammatory effects on the cells lining the walls of blood vessels. Adiponectin was first characterised in 1995 in differentiating 3T3-L1 adipocytes (Scherer PE et al.). In 1996 it was characterised in mice as the mRNA transcript most highly expressed in adipocytes (Maeda, 1996). In 2007, adiponectin was identified as a transcript highly expressed in preadipocytes (precursors of fat cells) differentiating into adipocytes.
High blood levels of adiponectin are associated with a reduced risk of heart attack. Low levels of adiponectin are found in people who are obese, and who are at increased risk of a heart attack.
Adipocytes produce and secrete a number of proteins, including leptin, adipsin, properdin, and tumor necrosis factor (TNF). An adipose tissue-specific factor was isolated and termed APM1 (AdiPose Most abundant gene transcript 1). The protein product of APM1 is adiponectin. The APM1 gene maps to chromosome 3q27.
Structure and Functions of Adiponectin
Adiponectin stimulation of its receptors, AdipoR1 and AdipoR2, increases the activities of 5′ AMP-activated protein kinase (AMPK) and peroxisome proliferator-activated receptor (PPAR), respectively, thereby contributing to healthy longevity as key anti-diabetic molecules. AdipoR1 and AdipoR2 were predicted to contain seven transmembrane helices with the opposite topology to G-protein-coupled receptors. There are four distinct regions of adiponectin. The first is a short signal sequence that targets the hormone for secretion outside the cell; next is a short region that varies between species; the third is a 65-amino acid region with similarity to collagenous proteins; the last is a globular domain. Overall this protein shows similarity to the complement 1Q factors (C1Q). However, when the 3-dimensional structure of the globular region was determined, a striking similarity to TNFα was observed, despite unrelated protein sequences.
Adiponectin automatically self-associates into larger structures. Initially, three adiponectin molecules bind together to form a homotrimer. The trimers continue to self-associate and form hexamers or dodecamers. Like the plasma concentration, the relative levels of the higher-order structures are sexually dimorphic, where females have increased proportions of the high-molecular weight forms. Recent studies showed that the high-molecular weight form may be the most biologically active form regarding glucose homeostasis. High-molecular-weight adiponectin was further found to be associated with a lower risk of diabetes with similar magnitude of association as total adiponectin. However, coronary artery disease has been found to be positively associated with high molecular weight adiponectin, but not with low molecular weight adiponectin.
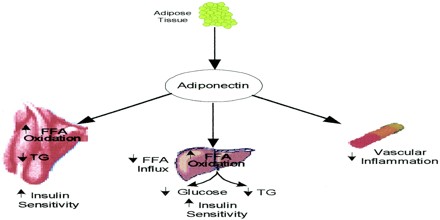
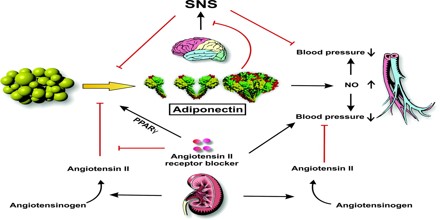
The physiological role of adiponectin has not yet been fully elucidated, but it is believed that it has the ability to reduce glucose, triglycerides, and free fatty acids and that it plays a major role in the pathogenesis of metabolic syndrome. Metabolic syndrome comprises a cluster of metabolic disorders that give rise to such metabolic risk factors as visceral obesity, insulin resistance, hyperglycaemia, dyslipidaemia, and hypertension. In addition, numerous experimental and clinical observations have shown decreased adiponectin bioactivity in obesity and obesity-related complications, including insulin resistance, diabetes, cardiovascular diseases, and non-alcoholic fatty liver disease (NAFLD).
Adiponectin is secreted into the bloodstream where it accounts for approximately 0.01% of all plasma protein at around 5-10 μg/mL (mg/L). In adults, plasma concentrations are higher in females than males, and are reduced in diabetics compared to non-diabetics. Weight reduction significantly increases circulating concentrations.
Regulation of Adiponectin Production
Though the relationship between plasma insulin and adiponectin levels has been studied extensively, the exact role of insulin in adiponectin biosynthesis and secretion remains contentious. In 3T3-L1 adipocytes, insulin has a direct stimulatory effect on adiponectin gene expression. Studies have also demonstrated the selectivity of insulin in adiponectin regulation and secretion.
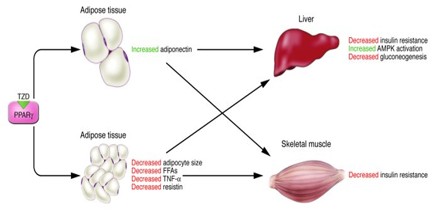
PPARγ is a member of the PPAR subfamilies of transcription factors, which is expressed mainly in adipose tissue and which is considered to be a positive regulator of adiponectin gene expression. Targeted deletion of PPARγ in adipose tissue of mice results in marked adipocyte hypocellularity and hypertrophy, elevated levels of plasma free fatty acids and triglyceride, and decreased levels of adiponectin.
Sterol-regulatory-element-binding proteins (SREBPs) are unique members of the basic helix-loop-helix leucine-zipper family of transcription factors. The binding of SREBPs to sterol response elements (SREs) promotes transcription and up-regulation of enzymes involved in lipid metabolism.
FoxO1 is a member of the forkhead box O transcription factor family. FoxO1 is involved in the regulation of adipocyte differentiation and positively regulates adiponectin transcription. NFAT comprises a family of transcription factors that have a definite role in 3T3-L1 adipocytes. NFATc4 and ATF3 negatively regulate adiponectin gene expression.