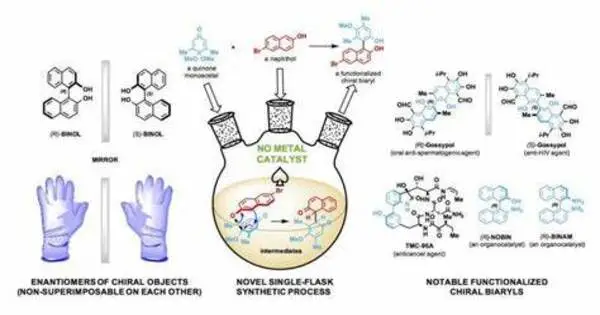
The discovery of innovative catalysts for the environmentally friendly manufacturing of fine chemicals and pharmaceuticals is an important topic of chemistry study. Green chemistry is concerned with developing more ecologically friendly and sustainable methods of carrying out chemical processes. Scientists have created a novel catalyst with a much smaller carbon footprint, paving the possibility for more environmentally friendly chemical and pharmaceutical manufacturing processes.
A team of researchers from the National University of Singapore (NUS), led by Associate Professor Lu Jiong from the Department of Chemistry in the NUS Faculty of Science, has developed a new class of catalysts known as heterogeneous geminal atom catalysts (GACs) that promotes greener and more sustainable manufacturing processes for fine chemicals and pharmaceuticals.
Fine chemical and pharmaceutical manufacturing are major contributors of air pollution, with recent research indicating that the pharmaceutical industry has a larger carbon footprint than the automotive industry. Aside from greenhouse gas emissions, the pharmaceutical business is also responsible for other major environmental consequences, such as water pollution caused by wastewater discharged by factories.
Developing alternative catalytic systems capable of achieving atomic-level precision while ensuring recoverability is at the forefront of our mission to revolutionize sustainable manufacturing processes for fine chemicals and pharmaceuticals. This ground-breaking achievement is the outcome of a close collaboration between several institutions.
Assoc Prof Lu
“Developing alternative catalytic systems capable of achieving atomic-level precision while ensuring recoverability is at the forefront of our mission to revolutionize sustainable manufacturing processes for fine chemicals and pharmaceuticals. This ground-breaking achievement is the outcome of a close collaboration between several institutions,” said Assoc Prof Lu.
The study was a collaboration involving Associate Professor Koh Ming Joo and Assistant Professor Zhu Ye from the Department of Chemistry under the NUS Faculty of Science, Professor Li Jun from Tsinghua University in China, Professor Javier Pérez-Ramírez from ETH Zurich in Switzerland, and Dr Xi Shibo from the Agency for Science, Technology and Research (A*STAR) in Singapore.
The research breakthrough was published in the scientific journal Nature.
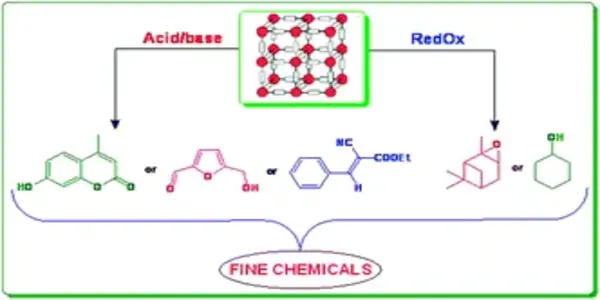
Developing a new class of catalyst
The synthesis of organic compounds requires a series of steps known as transition metal-catalyzed coupling reactions. These chemical reactions are indispensable for forming essential chemical bonds during the synthesis of a chemical compound. However, catalysts that are currently used in these reactions pose a number of challenges, such as high production cost, difficulty in catalyst separation for recovery and reuse, and metal contamination which is harmful to the environment. The structural architecture of current catalysts also limits their capacity to carry out complex reactions.
NUS researchers developed a new class of GACs with worldwide collaborators to overcome these problems and increase the possibilities for more sustainable and environmentally friendly pharmaceutical manufacturing procedures.
The inclusion of two metal cores comprised of copper ions, which allows for more efficient and selective reactions, is a major property of this new species of catalyst. The researchers employed a substance known as polymeric carbon nitride (PCN) as a supporting structure to hold the two copper ions in place so that they could work together in the chemical processes. The researchers tinkered with the structure until they discovered that 0.4 nanometres was the ideal spacing between these two copper ions for them to behave as a single unit in order to carry out key chemical reactions.
The innovative catalyst has a distinct heptazine chain structure that enables it to be dynamic and adaptive during chemical reactions enabling the two copper ions to efficiently bring two reactants together for the formation of a chemical bond; this chemical reaction is known as cross-coupling. This structure also lowers the minimal amount of energy required for a chemical reaction to take place.
The newly designed catalyst was then evaluated in several chemical reactions involved in the creation of commonly used pharmaceuticals and chemical compounds to demonstrate its effectiveness compared to conventional catalysts, and researchers quantified the environmental benefits of this new form of GACs.
Novel GACs for greener chemical processes
To showcase the versatility of the newly developed GACs, the researchers assessed their performance in various chemical reactions, such as the formation of multifunctional heterocyclic compounds that are commonly used in the production of pharmaceuticals.
The team also reported that the novel catalyst can increase the final product yield. For example, while utilizing the novel GACs, more bromide substrates were readily available, allowing the yield of dutasteride, which is generally used to treat prostate disease, to be increased from 53% to 62% when compared to conventional metal catalysts.
The catalyst was subjected to nine consecutive cycles of chemical reactions, and the researchers determined that it could remain stable with no apparent loss of copper ions from the original structure. This means that waste and the risk of metal contamination can be greatly reduced. Furthermore, the novel GACs are easily recovered and reused, emphasizing their potential to improve sustainability in the chemical and pharmaceutical industries.
The researchers also looked at the environmental benefits of employing the novel catalyst in chemical reactions and discovered that it had a carbon footprint that is ten times smaller than standard catalysts. This new class of GACs is an appealing choice for implementation in the fine chemical and pharmaceutical sectors because it outperforms conventional catalysts in cross-coupling reactions by increasing yield, efficiency, and environmental impact.
“Our goal in the near future is to create a library of GACs by carefully adjusting the specific types and combinations of geminal metal centers. This can potentially transform the conventional methods of chemical production. It could signify the dawn of a new era where GACs play a pivotal role in achieving greener and more environmentally friendly chemical and pharmaceutical manufacturing,” added Assoc Prof Lu.