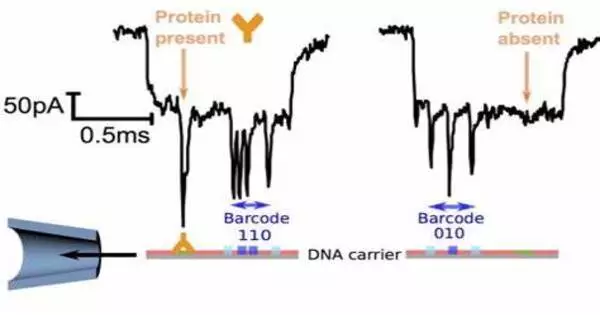
A nanoprobe with a barcode is a nanoscale probe or sensor that is equipped with a barcode for identification purposes. Nanoprobes are devices designed to interact with materials or biological systems at the nanoscale (one billionth of a meter). These probes can be used for a variety of purposes, such as medical diagnostics, imaging, and sensing.
Protein-splitting enzymes are essential in many physiological processes. In most cases, such proteases are inactive and only become active under certain conditions. Some are linked to diseases such as infections or cancer, making it critical to have methods for detecting active proteases. In the journal Angewandte Chemie, scientists have introduced a new class of protease-activity sensors: gold nanoparticles equipped with peptide DNA.
The team, led by Devleena Samanta and Anna Capasso (The University of Texas at Austin, USA), demonstrated that these nanoprobes can detect multiple active proteases in parallel (multiplexed measurement). The method operates at room temperature and does not necessitate complicated sample preparation or expensive instruments.
The team demonstrated the high sensitivity and selectivity of their new method by multiplexing detection of the proteases 3CL and caspase3. 3CL is a marker for active coronavirus infection, and COVID patients frequently have elevated caspase3 activity.
Devleena Samanta and Anna Capasso
Gold nanoparticles with peptide and DNA fragment chains are at the heart of the novel probes. The peptide structure is intended to be one that can be split by the protease being detected. The DNA serves as a unique barcode for identifying the peptide and also amplifies the signal. If the desired protease is present in active form in the sample, the peptide splits it. This releases the DNA barcode into the solution, where it can be detected based on its sequence.
The team uses a CRISPR/Cas12a test to detect this: the enzyme Cas12a is bound to a guide RNA (gRNA) to form an inactive complex. The gRNA contains a segment that specifically binds to the barcode DNA. This activates the Cas12a, allowing it to “cut up” single-stranded DNA (ssDNA). The researchers use ssDNA molecules with a fluorescing group (fluorophore) at one end and a quencher at the other, which “switches off” the fluorescence of the fluorophore (as long as they are close enough). When the ssDNA is cut up, the fluorophore and quencher move further apart. This produces strong fluorescence, indicating that the protease under test is present (detection limit of about 58 pM).
If no instruments are available on site and the test must go fast, detection is possible with the naked eye: if the protease splits the peptide on the probe, the surface charge of the gold nanoparticles changes and they aggregate. The color of these so-called “plasmonic nanostructures” depends significantly on their degree of aggregation. It is possible to detect nanomolar protease concentrations based on the color change in the test solution.
The team demonstrated the high sensitivity and selectivity of their new method by multiplexing detection of the proteases 3CL and caspase3. 3CL is a marker for active coronavirus infection, and COVID patients frequently have elevated caspase3 activity. The detection of cathepsin B, a protease associated with colorectal cancer, in three different tumor cell lines obtained from patients also demonstrated the clinical utility of this test.
When compared to commercial fluorescence-based protease sensors, these nanoprobes produce 100-fold higher fluorescence signals. Furthermore, almost any protease can be identified if the peptide it splits is known. Through multiplexing, these nanoprobes may be able to detect diseases earlier and improve the precision and reliability of diagnostic tests.