In studies headed by the University of Pennsylvania, Philadelphia, the protein cyclophilin A (CyPA) was silenced using siRNA to lessen tumor burden and prolong patient lives.
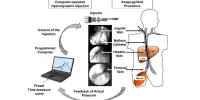
In the paper, “In vivo bone marrow microenvironment siRNA delivery using lipid-polymer nanoparticles for multiple myeloma therapy,” published in PNAS, the researchers detail a targeted nanoparticle platform to deliver nucleic acid therapeutics to bone marrow endothelial cells with a therapeutic payload.
A blood cancer called multiple myeloma (MM) can develop tumors outside of the bone marrow in the body’s organs (extramedullary illness). With unavoidable relapse following therapy and typically short life rates of 3 to 6 months for individuals with relapse as they develop resistance to the medicines, MM is currently treatable but incurable.
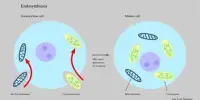
It is believed that endothelial cells in the bone marrow microenvironment are essential. Specifically, progression, survival, and chemotherapy resistance are mediated by the protein cyclophilin A (CyPA), which is released by bone marrow endothelial cells.
Although it can be difficult to deliver inhibitory compounds into the bone marrow endothelium, blocking CyPA could simultaneously slow the course of MM and make it more susceptible to chemotherapeutics.
Small interfering RNA (siRNA) treatments are excellent candidates for CyPA silencing and have the potential to silence any targeted gene, but they are constrained by their instability in the circulation and their difficulty crossing cell membranes.
The researchers went to a nanoparticle delivery system because they required a technique to deliver the possible medicine to the right place. To enable nucleic acid encapsulation, the scientists created nanoparticles made of a polymer-lipid hybrid material and a lipid-polyethylene glycol (PEG).
Through the functionalization of the nanoparticle surface chemistry, the nanoparticles were able to deliver the payload siRNA to particular tissues while reducing the RNA’s destruction by blood-borne enzymes.
The CyPA silencing siRNA payload and the nanoparticle delivery platform were used successfully in a living mouse model. CyPA’s silence reduced the invasion of multiple myeloma over bone marrow endothelial cells and interfered with interactions. CyPA knockdown increased sensitivity of cancer cells to bortezomib therapy, which decreased angiogenesis and proliferation and ultimately increased mice survival.
According to the authors, a broad enabling technology for delivering nucleic acid therapies to additional malignancies may be provided by this nanoparticle platform.