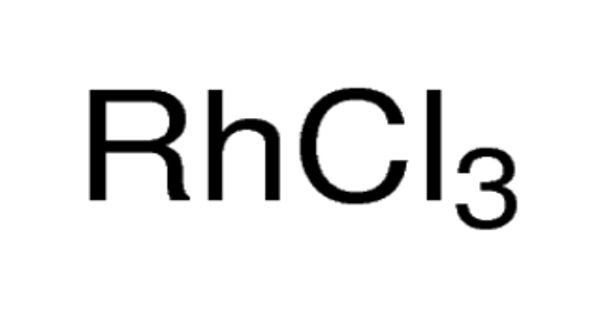Rhodium (III) chloride usually refers to hydrated rhodium trichloride, a molecular compound with the formula RhCl(H2O)3. It refers to inorganic compounds with the formula RhCl3(H2O)n, where n varies from 0 to 3. These are diamagnetic solids featuring octahedral Rh(III) centers. Depending on the value of n, the material is either a dense brown solid or a soluble reddish salt. The soluble trihydrated (n = 3) salt is widely used to prepare compounds used in homogeneous catalysis, notably for the industrial production of acetic acid and hydroformylation.
Rhodium (III) chloride precursor may be used to dope graphene with Rh nanoparticles to prepare hybrids for electrocatalysis in energy, sensing, and biomedical sectors.
Structures
Aqueous solutions of RhCl3(H2O)3 have been characterized by 103Rh NMR spectroscopy, which shows the presence of several species. Rhodium Trichloride Hydrate is an excellent water-soluble crystalline Rhodium source for uses compatible with chlorides. Hydrate or anhydrous forms may be purchased. Chloride compounds can conduct electricity when fused or dissolved in water. Chloride materials can be decomposed by electrolysis to chlorine gas and the metal. The proportions of which change with time and depend on the concentration of chloride. The relative distribution of these species determines the color of the solutions, which can range from yellow (the hexaaquo ion) to “raspberry-red”. Rhodium Trichloride Trihydrate is an excellent water-soluble crystalline Rhodium source for uses compatible with chlorides.
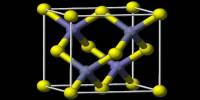
Rhodium (III) chlorides are the products of the separation of rhodium from the other platinum group metals. Anhydrous rhodium chloride crystallizes in the YCl3 and AlCl3 motif. The metal centers are octahedral, and the halides are doubly bridging. It is a dense brown solid that is insoluble in common solvents and of little value in the laboratory.
Preparation
RhCl3(H2O)3 is produced from salts such as Na3RhCl6, the latter being obtained in the purification of rhodium from the other platinum group metals such as platinum and iridium. It is produced by the action of hydrochloric acid on hydrated rhodium(III) oxide. The sodium salt is converted to H3RhCl6 by ion-exchange chromatography. Recrystallization of this acidic salt from water affords the hydrated trichloride, sometimes called “soluble rhodium trichloride.”
RhCl3(H2O)3 can be crystallized from a solution in concentrated hydrochloric acid. This method helps to remove nitrogen-containing impurities. Anhydrous RhCl3 is prepared by reaction of chlorine with rhodium sponge metal at 200–300°C. Above 800°C, the anhydrous chloride reverts to Rh metal and chlorine.
Application
Rhodium (III) chloride hydrate is used as a catalyst to prepare acetic acid. It is also used as a catalyst for reduction, oxidation of alkenes, hydration of acetylene, and isomerization of alkenes. It is involved in the hydrosilylation of alfa,beta-unsaturated esters to form dimethyl ketone trimethylsilyl acetals.