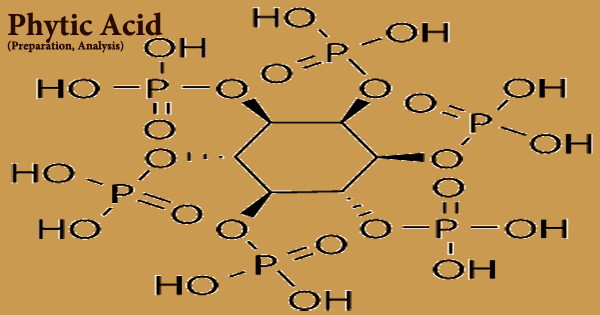
Phytic acid (PA), also known as inositol hexakisphosphate, hexaphosphoinositol, myo-inositol hexakisphosphate, IP6, is a light yellow to pale brown slurry liquid that is soluble in water, ethanol, and acetone but practically insoluble in ether, benzene, and chloroform. The phosphates are partly ionized at physiological pH, resulting in the phytate anion. It’s stable and doesn’t react well with powerful oxidizers.
The (myo) phytate anion is a colorless species that play an important nutritional role in many plant tissues, particularly bran and seeds, as the primary phosphorus storage form. It may also be found in a variety of legumes, cereals, and grains. It’s essentially non-toxic and should be stored in a cold, dry environment. General chemical standards can be followed for storage and shipment. Phytic acid (C6H18O24P6), which is found mostly in cereal grains and legumes, can form complexes with certain minerals, rendering them insoluble and hence indigestible.
The dietary minerals calcium, iron, and zinc have a significant binding affinity for phytic acid and phytate, which inhibits their absorption in the small intestine. PA is a naturally occurring chemical that forms during the maturity of seeds and cereal grains. It was originally discovered in 1855. It’s phosphorus in its storage form, which is a vital mineral for energy generation and the development of structural elements like cell membranes. Inositol esters containing less than six phosphates, such as inositol penta- (IP5), tetra- (IP4), and triphosphate, are among the lowest inositol polyphosphates (IP3). These are found in nature as phytic acid catabolites.

Because spinach also contains a lot of oxalic acids, only a tiny proportion of the calcium in it gets absorbed. It is structurally integrated with the protein bodies as phytin, mixed potassium, magnesium, and a calcium salt of inositol, and accounts for roughly 70% of the phosphate content in legume seeds. Non-ruminant animals do not have the enzyme phytase, which is necessary to hydrolyze the inositol-phosphate bonds, therefore phosphorus and inositol in phytate form are not accessible.
Because phytate from certain grains and beans cannot be digested, the unabsorbed phytate travels through the gastrointestinal system, increasing the phosphorus content of the excrement. In plants, PA is the most prevalent form of phosphorus. Endogenous phytases, which are present in most PA-containing seeds from higher plants, can partly dephosphorylate inositol hexaphosphate to generate breakdown products such as penta-, tetra-, and triphosphate during food processing and digestion.
In addition, viable low-phytic acid mutant lines have been produced in various crop species, with significantly lower phytic acid levels and accompanying increases in inorganic phosphorus in the seeds. In each experiment, the goal of chemical synthesis was to generate the required product by heating inosite and orthophosphoric acid together. Agricultural and sideline materials such as rice bran and plant germ are commonly used to extract PA. Solvent extraction aided technique, microwave-assisted extraction method, ultrasonic-assisted extraction method, membrane separation assist method, and others are the most common extraction methods nowadays.
Phytic acid and its metabolites play numerous vital roles for seedling plants, despite the fact that they are indigestible for many animals since they are found in seeds and grains. Another technique for preparing phytic acid is to utilize an anion exchange resin to adsorb the phytic acid in the extract, and then to elute the adsorbed phytic acid with an eluent to get phytate. Phytic acid serves as phosphorus storage, an energy store, a source of cations, and a source of myo-inositol, among other things (a cell wall precursor).
In vegetables like scallion and cabbage leaves, as well as fruits including apples, oranges, bananas, and pears, no measurable phytate (less than 0.02 percent of wet weight) was found. The principal technique of separation and quantification is high-performance liquid chromatography (HPLC). Phytic acid and inositol phosphates can be separated as distinct substances using HPLC. The dietary minerals calcium, iron, and zinc have a significant affinity for phytic acid, which inhibits their absorption from the small intestine.
The chemicals employed in this technique, however, must be clean and devoid of metals, otherwise, the data would be distorted. Polyphenols and tannins, for example, are phytochemicals that impact the binding. When iron and zinc bond to phytic acid, they produce insoluble precipitates that make the intestines less absorbable. While phytic acid binds to or chelates essential minerals, it also inhibits enzymes that help us digest our food, such as pepsin, which helps the stomach break down proteins, and amylase, which helps the body break down starch into sugar.
“Dephytinization should be regarded as a significant method to enhance iron feeding during the weaning phase,” says the study, because phytic acid might impair iron absorption. PAs have been proposed to operate as neuromodulators, whilst naturally occurring chemicals have been hypothesized to act as second messengers. PA is also thought to be a natural antioxidant with the ability to reduce lipid peroxidation.
Information Sources: